
January 27, 2026
Compliance and Inspection Readiness in Pharmacovigilance
Learn how Fidelity builds compliant, inspection-ready pharmacovigilance systems through robust PV-IT design, documentation control, and regulatory alignment with EMA, FDA, and WHO expectations.
Every pharmacovigilance system exists under the constant expectation of inspection. Regulators must be able to verify that the data used to protect patients is accurate, traceable, and reproducible. Compliance and inspection readiness are, therefore, not final steps in pharmacovigilance but continuous disciplines that sustain its credibility.
When compliance becomes part of design, inspection becomes confirmation rather than confrontation. Fidelity Health Services structures its Pharmacovigilance Information Technology (PV-IT) frameworks around this principle, ensuring that every database, workflow, and report can withstand the closest regulatory scrutiny.

Defining Compliance in Pharmacovigilance
Compliance in pharmacovigilance means adherence to all applicable regulations, guidance documents, and internal quality standards governing the collection, processing, and reporting of safety data. It is both procedural and technical:
Procedural compliance requires validated processes, defined responsibilities, and updated standard operating procedures.
Technical compliance demands validated systems, secure data handling, and audit-ready documentation.
Regulatory expectations are framed by international guidelines such as the EMA Good Pharmacovigilance Practices, FDA 21 CFR Part 11, and ICH E2E. Each emphasises the same core concept: every safety activity must be documented, controlled, and verifiable.
The Continuous Nature of Inspection Readiness
Inspection readiness is not an event before an audit; it is a state of perpetual preparedness. A pharmacovigilance team that maintains validated systems, complete documentation, and trained staff is inspection-ready every day.
Inspection readiness ensures:
- Availability of traceable data for all cases.
- Version control of procedural and system documents.
- Clearly defined roles and training records for responsible personnel.
- Consistent evidence of quality oversight.
Fidelity’s PV-IT solutions embed these expectations within daily operations, reducing the need for reactive preparation when regulators announce an inspection.
Common Inspection Findings in Pharmacovigilance regulatory inspections often reveal recurring weaknesses:
- Incomplete or inconsistent documentation.
- Unvalidated or outdated safety databases.
- Lack of audit trails or data integrity breaches.
- Inadequate training and role definitions.
- Non-standard global operating procedures.
Each of these gaps stems from systems designed for output rather than compliance. Fidelity addresses them through architecture that treats compliance as a function, not a formality.
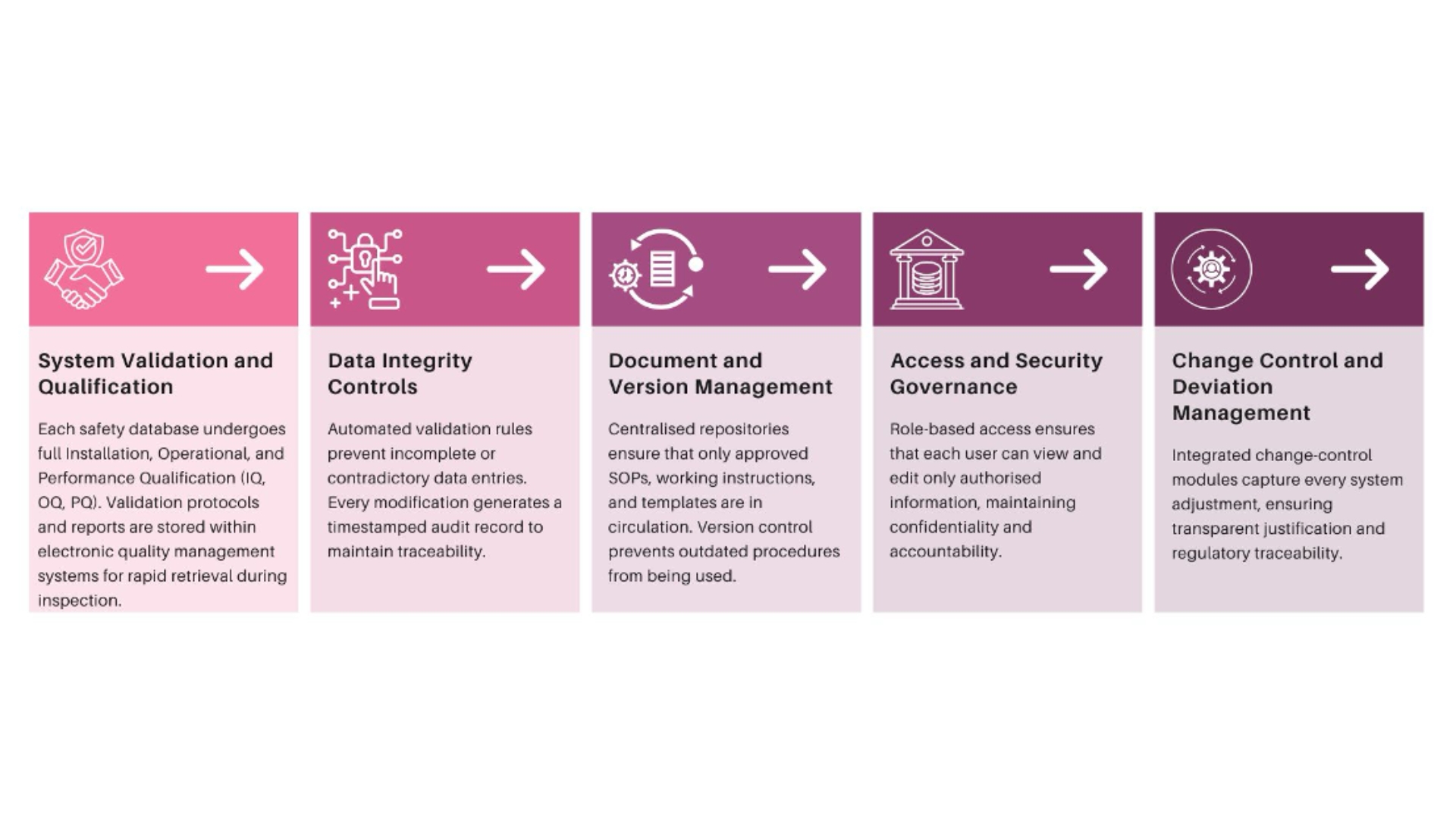
Building Compliance into PV-IT Architecture
Fidelity’s approach begins with system design. Every module from case intake to reporting includes embedded validation checks, audit capabilities, and security protocols.
Preparing for Regulatory Inspections
An inspection evaluates both systems and people. Readiness, therefore, requires coordination across functions. Fidelity supports clients through structured readiness programs that include:
- Mock Audits: Simulated inspections to evaluate preparedness and close gaps.
- Audit Response Protocols: Defined procedures for document retrieval and question handling.
- Training and Coaching: Preparing personnel to communicate effectively with inspectors.
- Evidence Mapping: Indexing of validation, training, and compliance records for quick access.
These activities create a culture where inspect: When Machines Start to Listen
Regulatory Expectations by Region: European Union (EMA)
Inspectors review system validation, case processing timelines, and compliance with GVP Modules I and IX.
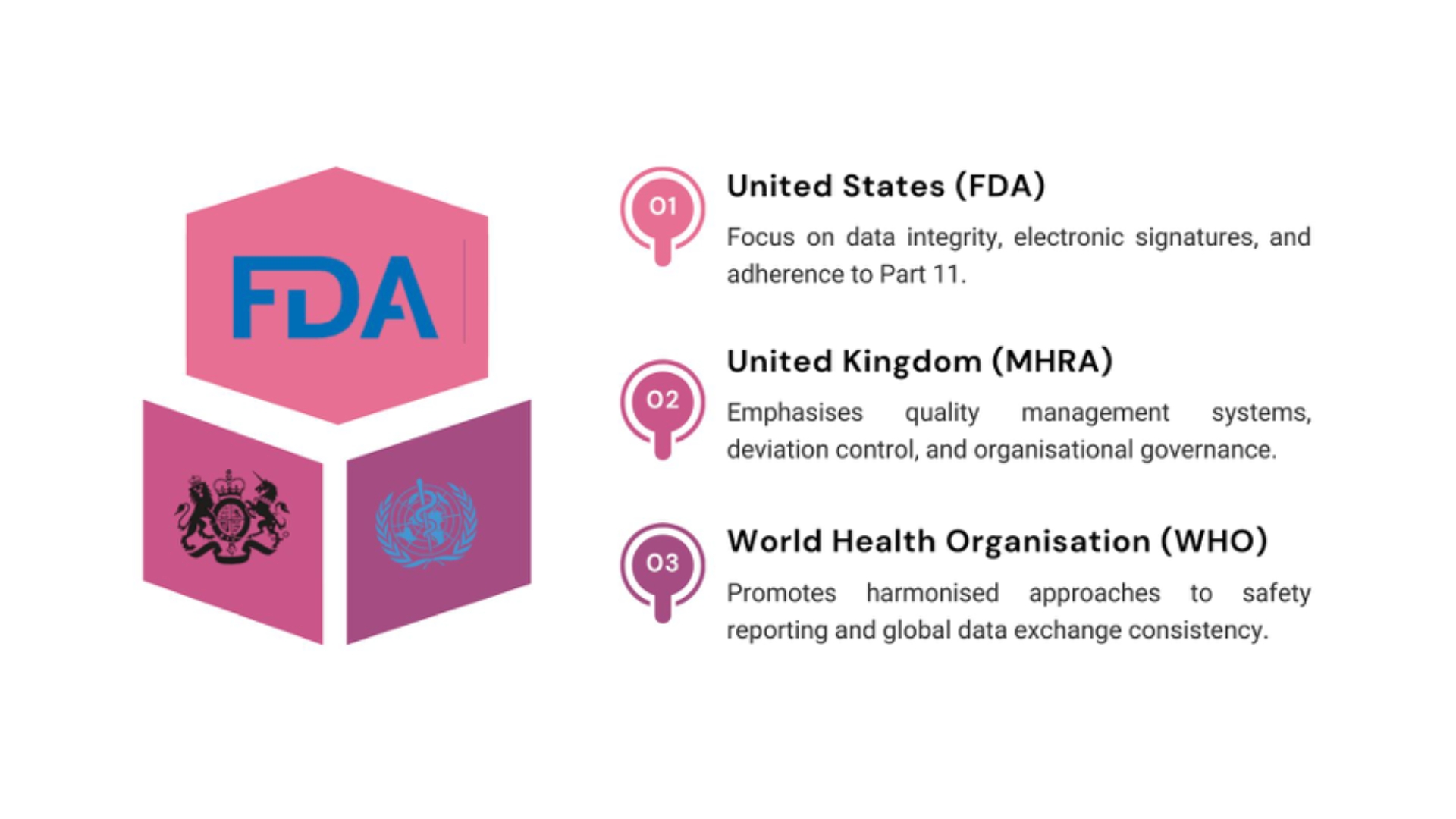
Fidelity’s global team aligns every PV-IT system with these frameworks, ensuring readiness for multi-agency audits.
Integration with Other PV-IT Components
Inspection readiness cannot function in isolation. It depends on:
-Validated data from Data Validation and Migration processes.
-Modern infrastructure from Safety System Modernisation.
Consistent procedures from Operational Harmonisation. Together, these create a closed-loop compliance ecosystem that supports inspection success.
Explore related articles:
Safety System Modernisation: Building the Infrastructure of Compliance
Operational Harmonisation: Aligning People, Process, and Performance
Risk Management in Pharmacovigilance: From Detection to Prevention
Fidelity’s Compliance and Inspection Readiness Framework
Fidelity’s framework operates on three pillars: Control, Documentation, and Confidence.
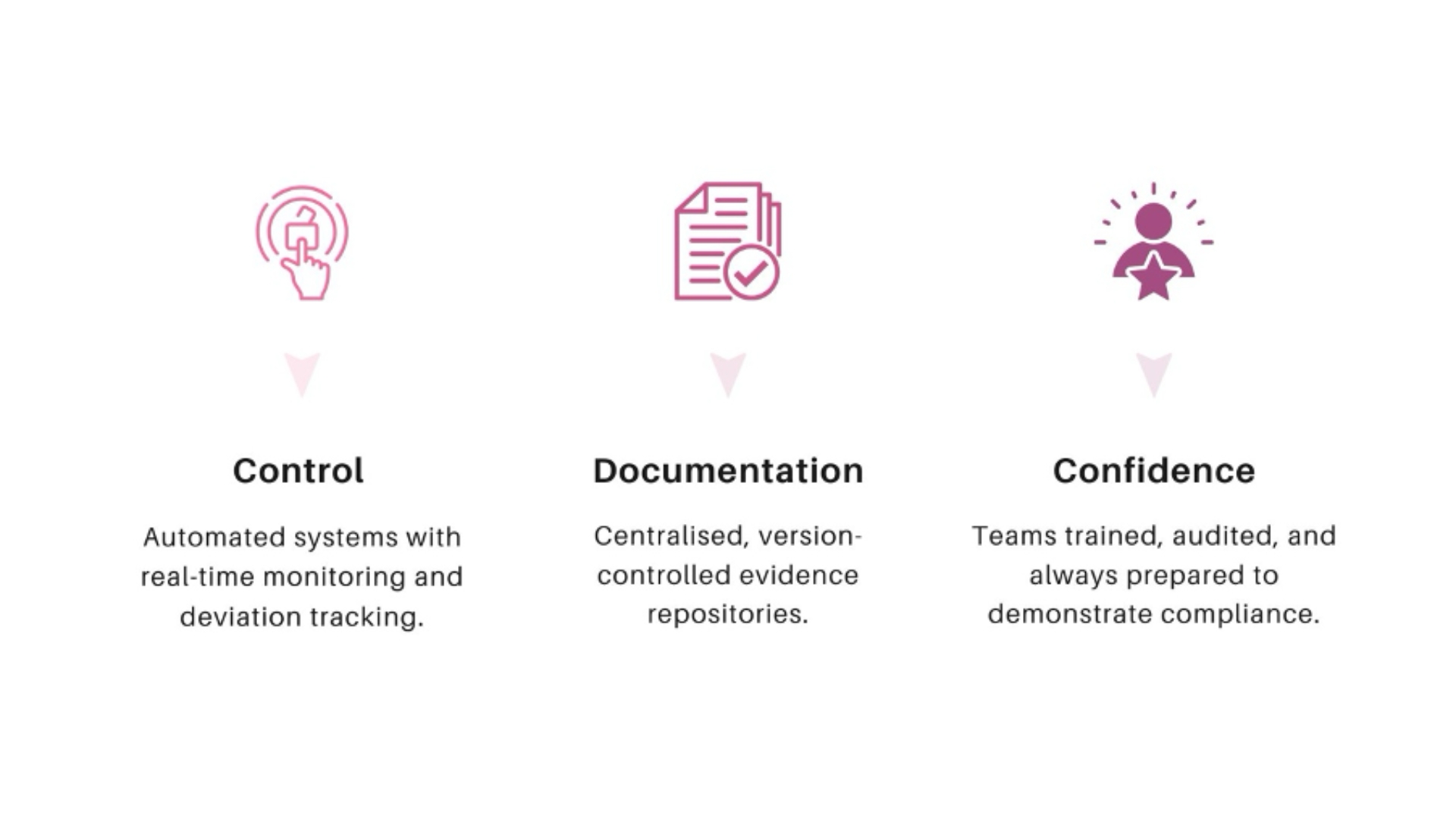
Through this model, Fidelity transforms compliance from a defensive measure into a competitive advantage.
Future of Digital Compliance
The next decade will witness the rise of continuous auditing through digital tools. AI-driven monitoring will identify anomalies in data flow instantly, allowing organisations to address issues before regulators do. Cloud-based compliance dashboards will integrate validation, quality metrics, and inspection readiness into a single visual platform.
Fidelity is actively developing PV-IT ecosystems that support these innovations. The vision is an environment where compliance is verified not periodically but continuously, powered by automation and guided by human oversight.
Conclusion
Inspection readiness reflects the maturity of an organisation’s commitment to patient safety. When built into systems, culture, and daily practice, it ensures that pharmacovigilance stands on evidence rather than assurance.
At Fidelity Health Services, every PV-IT implementation is designed for compliance from the start. Each module, dashboard, and workflow is engineered to meet regulatory scrutiny and to give organisations the confidence that comes only from readiness.
To complete the PV-IT series, continue to The Future of PV-IT Integration, Intelligence, and Integrity.