
December 30, 2025
Risk Management in Pharmacovigilance: From Detection to Prevention
Medicinal products are inherently associated with adverse reactions. These reactions vary in severity, likelihood of occurrence, impact on individual patients, and overall public health consequences.

At the time of initial marketing authorisation approval, it is not possible to identify or fully characterise all adverse reactions and risks associated with a medicine. Several important risks are only discovered and better understood during the post-authorisation phase, once the product is used in real-world clinical settings
This reality makes risk management a fundamental pillar of pharmacovigilance. Beyond regulatory obligation, effective risk management represents a continuous commitment to patient safety, benefit-risk optimisation, and regulatory confidence throughout the product lifecycle.
In today’s regulatory environment, pharmacovigilance has evolved from reactive case processing into a proactive, data-driven discipline. Risk is no longer something that is only documented; it is something that must be anticipated, monitored, minimised, and continuously reassessed.
The Foundation of Risk Management in Pharmacovigilance
Risk management in pharmacovigilance refers to the systematic identification, characterisation, prevention, and minimisation of risks associated with medicinal products across their entire lifecycle, from clinical development to post-marketing surveillance.
Regulatory authorities expect companies to demonstrate that risks are not only identified but actively controlled. This expectation is formalised through structured risk management frameworks such as Risk Management Plans (RMPs) in Europe and Risk Evaluation and Mitigation Strategies (REMS) in the United States.
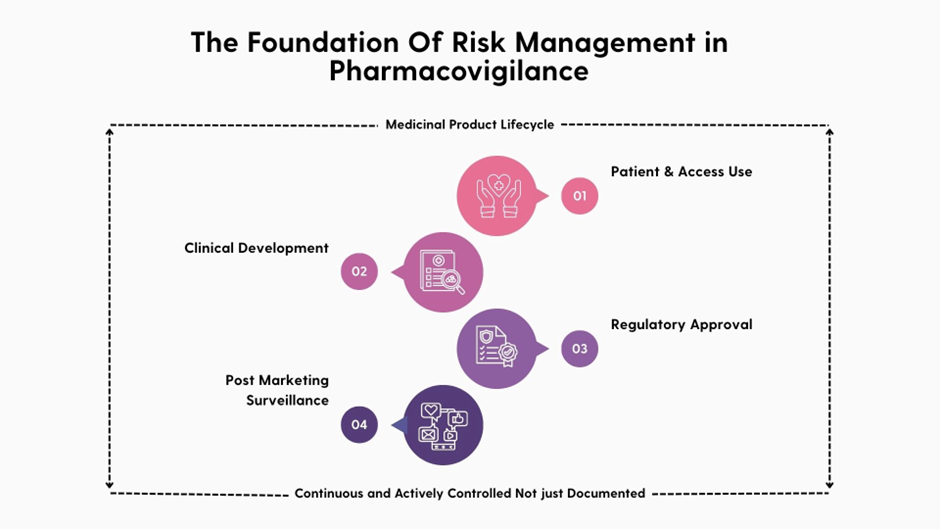
An RMP is a legal requirement for initial marketing authorisation applications in the European Union and remains mandatory throughout the post-authorisation phase of a medicine. Its purpose is to document known and potential risks, describe routine and additional risk minimisation measures, and outline plans for further characterisation of safety concerns.
Similarly, a REMS is a drug safety programme that the U.S. Food and Drug Administration may require for medicines with serious safety concerns. The objective of a REMS is to ensure that the benefits of a medication continue to outweigh its risks through targeted risk prevention and mitigation measures.
From Signal Detection to Strategic Decision-Making
Historically, signal detection was a largely retrospective process, relying on manual reviews, delayed reporting, and fragmented datasets. Modern pharmacovigilance systems have transformed this approach through integrated Pharmacovigilance Information Technology (PV-IT) platforms that enable continuous surveillance and structured decision-making.
Automated signal detection tools now analyse global safety databases using statistical methodologies to identify unusual reporting patterns. Once a potential signal is detected, it enters a controlled workflow for medical assessment, benefit-risk evaluation, and regulatory decision-making.
This structured approach allows organisations to manage increasing case volumes while maintaining compliance with regulatory authorities such as the European Medicines Agency and the U.S. Food and Drug Administration.
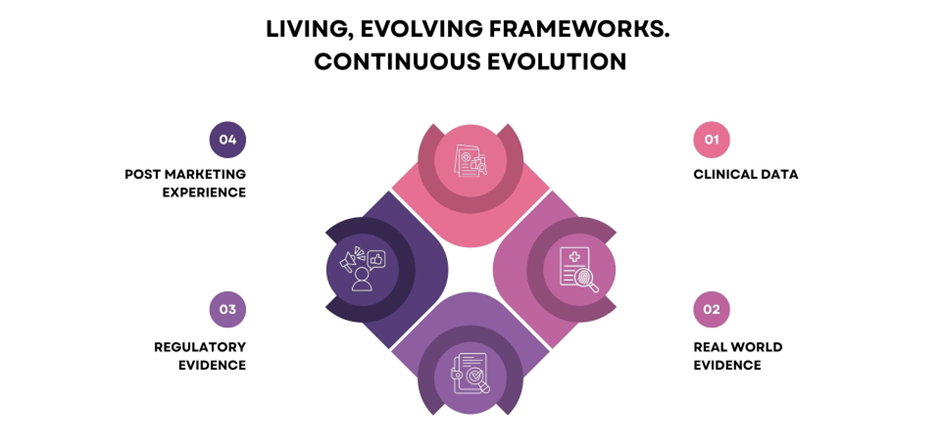
The Role of Risk Management Plans (RMPs) and REMS
Risk Management Plans and REMS are not static documents. They are living frameworks that evolve with accumulating clinical data, post-marketing experience, real-world evidence, and regulatory feedback.
REMS programmes focus on preventing, monitoring, and managing specific serious risks associated with a medicine. These programmes may involve informing and educating healthcare professionals and patients, reinforcing safe-use behaviours, and implementing controls to reduce the frequency or severity of adverse events.
Risk minimisation activities may include prescriber training, patient education materials, controlled distribution systems, monitoring programmes, and ongoing assessment of the effectiveness of these measures.
Through structured governance, version control, and traceable documentation, RMPs and REMS provide regulators with assurance that risks are actively managed and that patient safety remains central to commercial decision-making.
Fidelity’s Support for RMP and REMS Management
Fidelity Health Services supports Marketing Authorisation Holders globally in the end-to-end management of RMPs and REMS, including the execution of product-specific post-authorisation commitments across regions. Our support spans both strategic and operational dimensions, ensuring that risk management obligations are consistently implemented, effectively monitored, and maintained in a state of continuous readiness for inspection.
Fidelity’s capabilities include the preparation of new RMPs for a wide range of product types, including biosimilar products, hybrid products, generic medicines, informed consent products, well-established medicinal use products, full marketing authorisation applications, and advanced therapy medicinal products. These RMPs are developed and updated in line with evolving regulatory expectations.
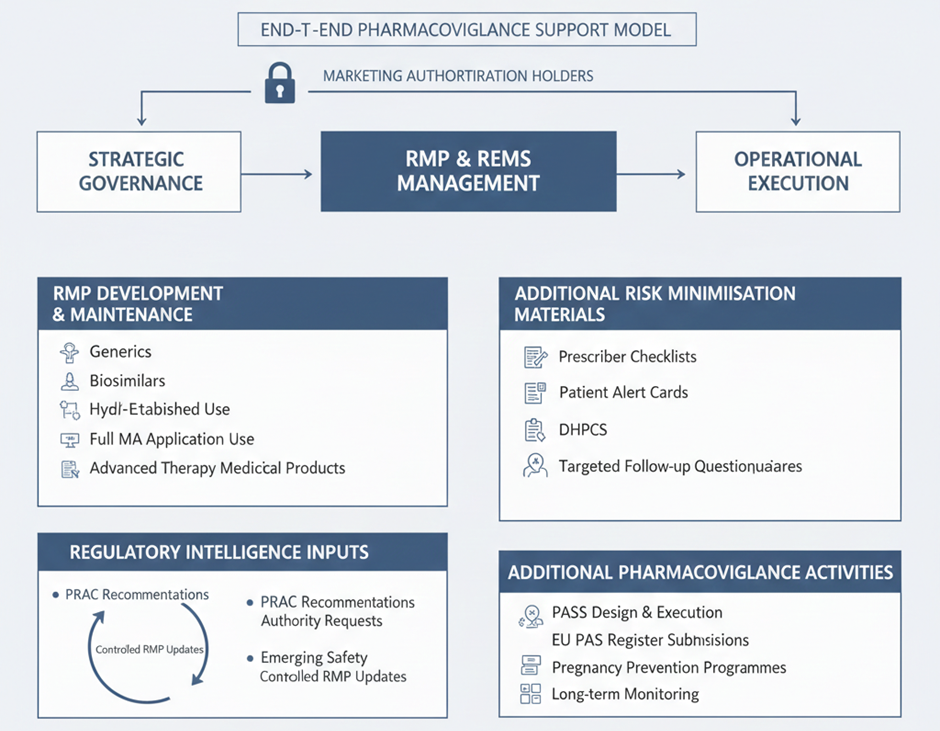
We continuously track and identify regulatory updates that may impact approved RMPs, including PRAC recommendations, competent authority requests, and emerging safety information that may alter the benefit-risk profile of a medicine. Where changes in benefit-risk balance are identified, Fidelity supports timely updates to RMPs to ensure continued regulatory alignment.
Fidelity also develops and maintains Targeted Follow-up Questionnaires and Additional Risk Minimisation Materials such as prescriber checklists, patient alert cards, healthcare professional and patient guides, prescriber brochures, Direct Healthcare Professional Communications, and training materials.
In addition, we support the design and execution of Additional Pharmacovigilance activities, including Post-Authorisation Safety Studies, pregnancy prevention programmes for teratogenic medicines, and registration of PASS studies in the EU PAS Register. Where required, Fidelity supports participation in pregnancy registries and long-term monitoring programmes.
The effectiveness of additional risk minimisation measures and additional pharmacovigilance activities is tracked using validated process and outcome indicators, providing measurable evidence of compliance and real-world impact.
Regulatory Inspection Risk and Operational Challenges
Achieving and sustaining compliance with RMP and REMS commitments is extremely challenging for both global and local pharmaceutical and healthcare organisations. The complexity increases further when managing multiple products across regions with differing regulatory expectations.
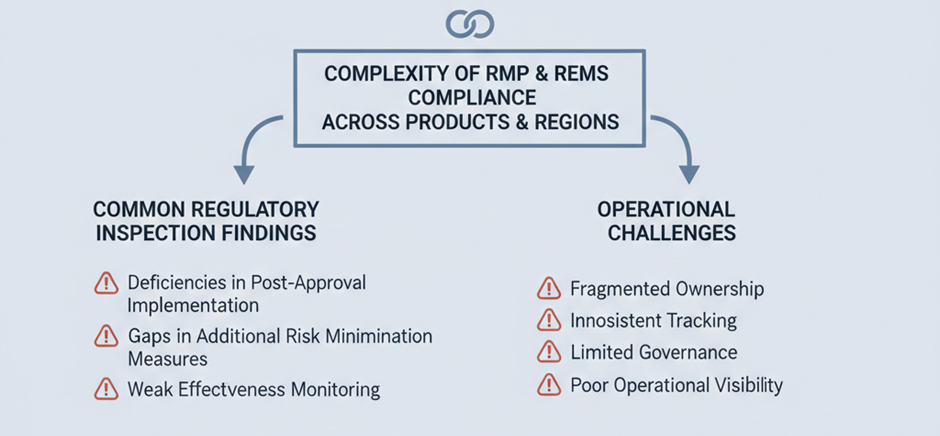
Regulatory inspections frequently identify critical or major observations related to deficiencies in the implementation and oversight of post-approval risk management commitments. In particular, gaps in the execution and effectiveness monitoring of additional risk minimisation measures and additional pharmacovigilance activities remain a common inspection finding.
To avoid such observations, it is essential to collaborate with a seasoned and trustworthy service provider that understands both regulatory expectations and operational realities.
Integrating Risk Management within PV-IT Systems
At Fidelity Health Services, risk management is embedded within a broader PV-IT ecosystem rather than treated as a standalone activity. Our integrated framework connects validated safety databases, automated signal detection, workflow automation, and regulatory reporting into a single, traceable environment.
This integration ensures that safety data, risk evaluation, and regulatory actions are aligned in real time. Decision-making becomes faster, more transparent, and fully auditable, supporting both day-to-day operations and regulatory inspections.
The Future of Risk Management in Pharmacovigilance

The future of pharmacovigilance risk management lies in predictive analytics, intelligent automation, and real-time global collaboration. Advanced analytics and artificial intelligence will increasingly support earlier identification of safety trends, allowing risks to be mitigated before they translate into patient harm or regulatory findings.Fidelity’s approach ensures that technological advancement is always balanced with scientific judgement, regulatory compliance, and patient-centric decision-making.
Conclusion
Risk management defines the maturity of a pharmacovigilance system. When implemented effectively, it transforms pharmacovigilance from a reactive function into a preventive, strategic discipline.
Through structured RMP and REMS management, robust PV-IT integration, and inspection-ready execution of post-authorisation commitments, Fidelity Health Services helps organisations protect patients, maintain regulatory confidence, and sustain long-term product success.