
January 12, 2026
Technology Enablement in PV: When Machines Start to Listen
Discover how technology enablement is redefining pharmacovigilance. Learn how Fidelity integrates automation, AI, and analytics within PV-IT to create intelligent, compliant safety systems.
For decades, pharmacovigilance relied on observation. Humans listened to reports, reviewed narratives, and made decisions based on clinical expertise. Today, that act of listening has been extended to technology. Systems can now recognise medical terms, detect anomalies, and even predict potential risks before they occur. This transformation, known as technology enablement, has redefined how global organisations protect patient safety.
At Fidelity Health Services, technology enablement within Pharmacovigilance Information Technology (PV-IT) represents the fusion of automation, analytics, and human oversight. It allows machines to process what once required hours of manual work, while experts focus on interpretation and strategy. The result is a pharmacovigilance environment that is faster, smarter, and always inspection-ready.

What Technology Enablement Means in PV-IT
Technology enablement is the process of embedding intelligent tools into every stage of pharmacovigilance. It enhances accuracy, scalability, and compliance by integrating digital solutions into case intake, assessment, and reporting.
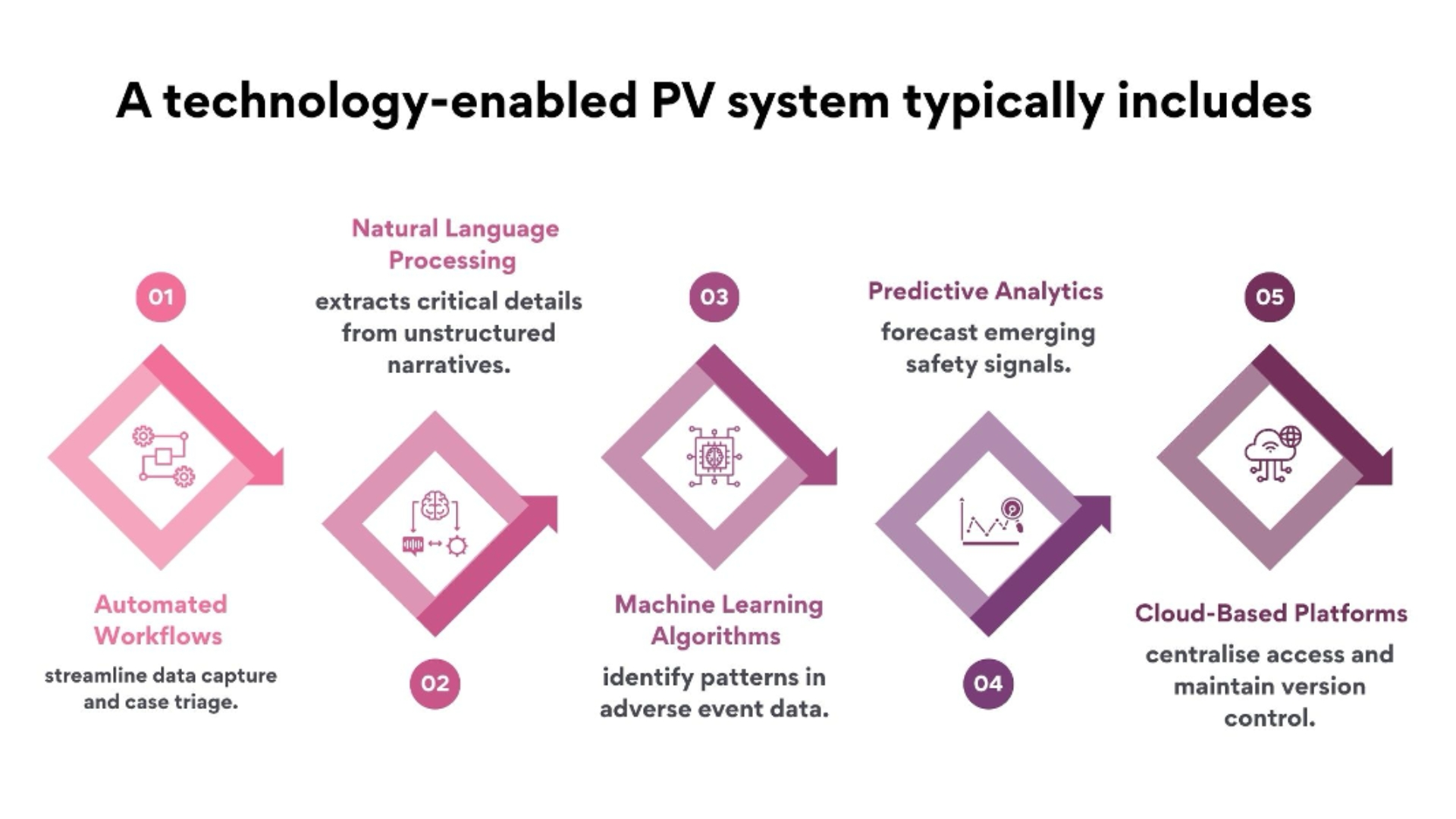
These elements create a digital nervous system where data moves seamlessly, decisions are supported by evidence, and compliance is embedded by design.
From Automation to Intelligence
Early PV technologies were built to automate repetitive tasks. Modern systems go further; they interpret context. Through artificial intelligence, safety databases can now differentiate between medically meaningful and duplicate cases. They can classify seriousness, extract dates, and flag inconsistencies automatically.
This progression from automation to intelligence has transformed pharmacovigilance from a reactive process to a predictive discipline. Machines do not replace human judgment; they refine it by presenting structured, validated insights drawn from vast data sets.
Related Reading: Risk Management in Pharmacovigilance: From Detection to Prevention
Core Technologies Powering PV Transformation
- Natural Language Processing (NLP)
- Robotic Process Automation (RPA)
- Artificial Intelligence and Machine Learning (AI/ML)
- Advanced Analytics and Visualisation
- Cloud and API (Application Programming Interface) Integration
NLP enables systems to read and interpret narrative data from emails, call notes, or medical literature. It converts language into structured information, drastically reducing manual transcription errors.
RPA handles repetitive, rule-based tasks such as data entry, reconciliation, and follow-up scheduling. It ensures consistency and frees personnel for analytical functions.
AI/ML algorithms identify subtle correlations and patterns that may indicate emerging safety issues. They also support risk scoring and benefit-risk evaluation.
Interactive dashboards provide real-time visibility into workflow efficiency, regulatory timelines, and safety trends, supporting evidence-based decision-making.
Cloud architecture ensures scalability and security, while API (Application Programming Interface) connectivity allows seamless integration between safety systems, regulatory portals, and data warehouses.
Together, these technologies create a unified environment that strengthens both operational efficiency and scientific rigour.

Fidelity’s Approach to Technology Enablement
Fidelity Health Services applies technology enablement as a structured, validated process rather than a standalone upgrade. Every deployment follows a life-cycle approach aligned with EMA, FDA, and ICH guidelines.
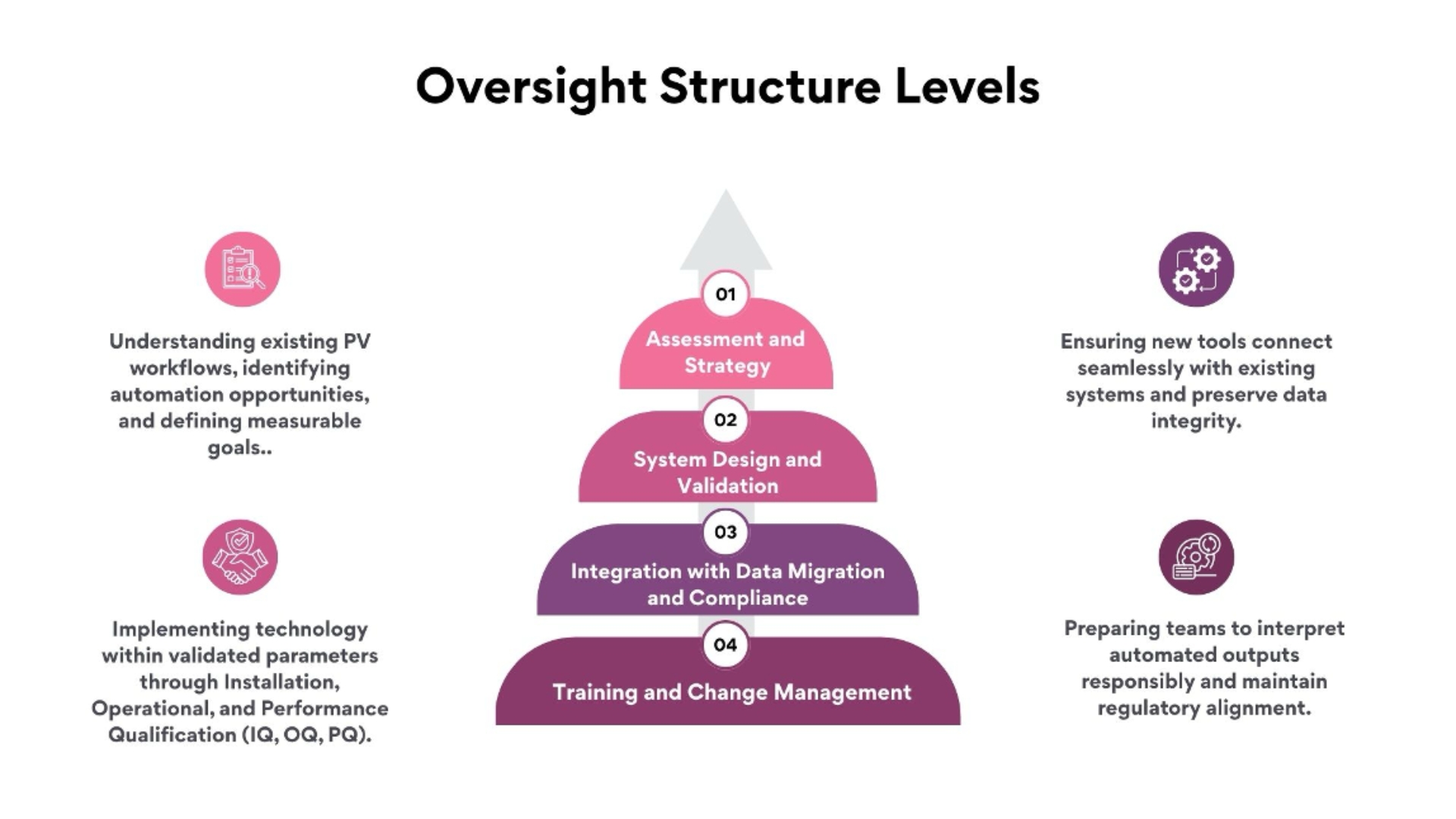
This structured model ensures that every innovation enhances accuracy without compromising compliance.
Explore related topics: Safety System Modernisation, Building the Infrastructure of Compliance and Data Validation and Migration, The Hidden Engines of Trust in Pharmacovigilance.
Regulatory Perspective on Digital Transformation
Global health authorities recognise the value of automation but continue to emphasise validation and oversight.
- The EMA requires technology implementations to meet Good Automated Manufacturing Practice (GAMP 5) principles.
- The FDA expects full documentation of electronic records under 21 CFR Part 11.
- The WHO UMC encourages the use of data science to strengthen signal detection, but within auditable frameworks.
Fidelity aligns every PV-IT deployment with these expectations, ensuring that innovation operates within regulatory boundaries.
Benefits of Technology-Enabled Pharmacovigilance
- Speed and Efficiency - Automation accelerates case processing and submission timelines.
- Accuracy - Validated systems eliminate transcription and reporting errors.
- Scalability - Cloud-based platforms support multi-region operations without added complexity.
- Transparency - Central dashboards provide continuous visibility into performance metrics.
- Compliance Continuity - Built-in audit trails guarantee inspection readiness at all times.
Technology enablement converts pharmacovigilance from a compliance function into a strategic capability that delivers measurable value.
Challenges in Digital Adoption
Despite its advantages, technology enablement introduces new responsibilities.
- System Validation Complexity - Every automated function must be validated to meet GxP standards.
- Data Security - Cloud systems demand robust encryption and access controls.
- Cultural Adaptation - Teams must learn to trust machine-generated insights.
Fidelity mitigates these challenges through rigorous validation documentation, secure architecture, and tailored user training. Technology adoption becomes a guided journey rather than a disruption.

The Future of Listening Machines
The future of pharmacovigilance belongs to systems that not only process data but also interpret emotion and urgency within reports. Advances in semantic AI and voice analytics will allow technology to understand patient language, detect distress, and prioritise cases dynamically.
In the coming years, the partnership between human expertise and intelligent systems will define global drug safety. Machines will listen, analyse, and predict, but humans will continue to lead with judgment, ethics, and empathy.
Fidelity’s mission is to design PV-IT ecosystems that enable this collaboration, where automation supports decision-making and technology preserves the human essence of safety science.
Conclusion
Technology enablement has transformed pharmacovigilance into a living network of data, algorithms, and insight. It allows safety systems to think faster, act earlier, and operate with absolute consistency.
At Fidelity Health Services, technology does more than automate processes. It listens, learns, and strengthens the bridge between science and compliance. Through structured PV-IT enablement, Fidelity ensures that every safety system evolves with innovation while remaining rooted in trust.
Continue exploring the PV-IT series with Compliance and Inspection Readiness in Pharmacovigilance and The Future of PV-IT Integration, Intelligence, and Integrity.