
January 29, 2026
Silent Compliance Risks in Pharmacovigilance Nobody Notices Until Inspection
Most pharmacovigilance inspection findings do not result from missing adverse events or late submissions. They originate from silent compliance risks. These risks remain invisible during daily operations but surface immediately when regulators begin asking how data moved, who controlled it, and where decisions were documented.
Organisations often believe they are compliant because cases are processed on time and reports are submitted successfully. Inspections, however, evaluate something different. Regulators assess whether systems can explain themselves. When explanations rely on assumptions rather than evidence, findings follow.

What Silent Compliance Risks Really Are?
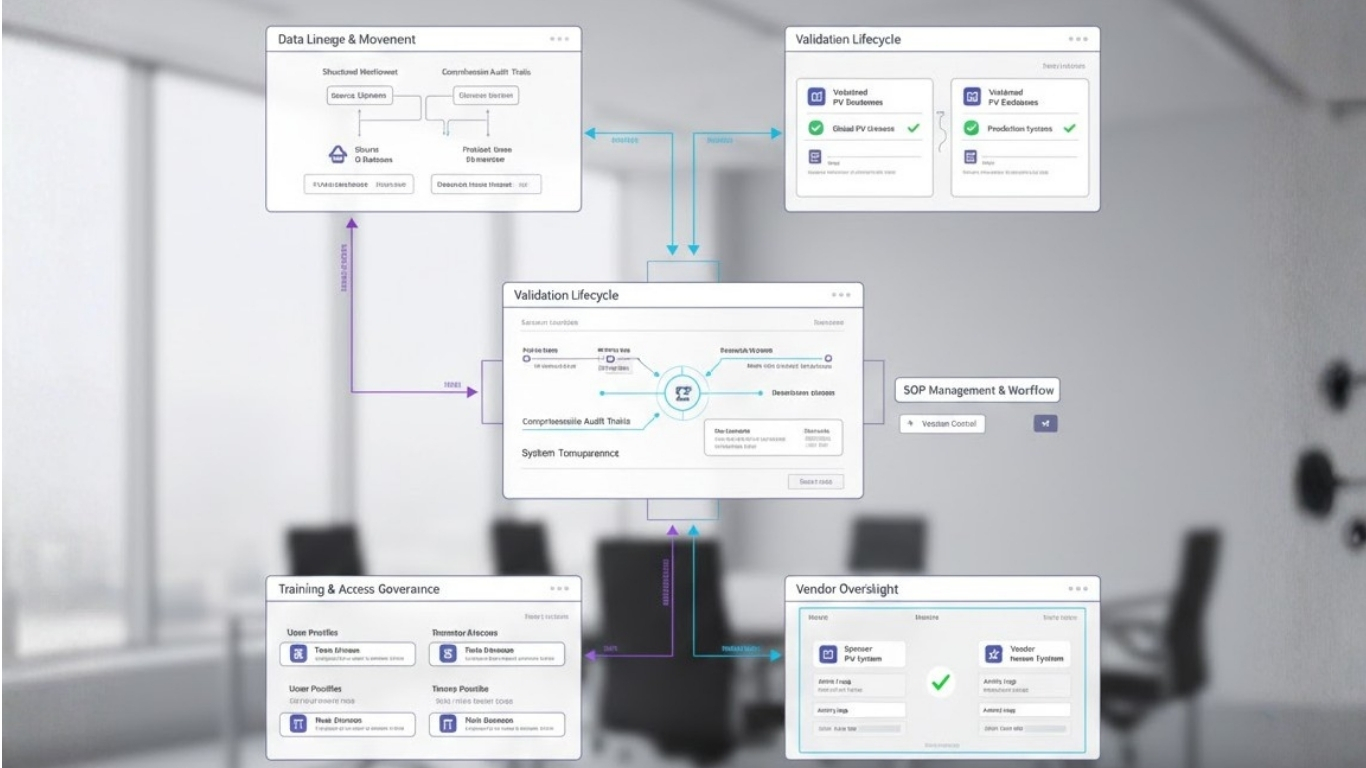
Silent compliance risks are weaknesses embedded within systems, processes, or governance structures that do not interrupt routine workflows but undermine inspection readiness. They are created when operational efficiency is prioritised without equal emphasis on traceability, validation, and documentation.
These risks accumulate slowly. They are rarely flagged by metrics or dashboards. Yet once inspectors begin tracing a single case or system change, the absence of controlled evidence becomes visible.
Where Inspectors Most Often Find Hidden Gaps?
One of the most common inspection issues involves data that cannot clearly demonstrate its journey. Safety data may be accurate, but if its movement between systems, vendors, or versions cannot be reconstructed, regulators consider the control insufficient. Questions about where data originated, how it was transformed, and what checks governed each step must be answerable without interpretation.
Another frequent finding involves validation that exists historically but cannot be aligned with the current system state. Validation performed during implementation loses value if subsequent changes are not governed through controlled change management. Inspectors from the European Medicines Agency and the U.S. Food and Drug Administration consistently assess whether validation evidence reflects the live environment, not a past configuration.
Procedural misalignment is another silent risk. Standard operating procedures often describe intended workflows rather than actual system behaviour. Over time, automation, vendor processes, or informal practices diverge from documented procedures. During inspection, this disconnect is interpreted as a lack of procedural control, even when teams perform competently.
Training and access governance also create hidden exposure. When user permissions are not continuously aligned with training completion and role changes, organisations struggle to demonstrate that only qualified individuals perform regulated activities. This issue rarely disrupts operations but frequently appears as a formal finding.
Vendor oversight presents one of the most underestimated risks. While quality agreements and periodic reviews exist, inspectors focus on whether sponsors retain real-time visibility and control. If vendor actions are not fully traceable within the sponsor’s PV environment, accountability gaps emerge.

Why These Risks Go Unnoticed Internally?
Silent compliance risks persist because internal reviews tend to focus on outputs rather than explainability. Metrics prioritise volume and timelines. Systems are designed to process cases efficiently, not to narrate their own compliance history.
As organisations scale, complexity increases faster than governance. Each system change, vendor addition, or geographic expansion introduces small inconsistencies that compound over time. Without intentional design for inspection logic, compliance debt grows quietly.
Preventing Silent Risks Through PV-IT Design
At Fidelity Health Services, compliance is treated as a system property rather than a periodic exercise. PV-IT environments are designed to ensure that every action generates traceable, reconstructable evidence.
This approach ensures that
- Data movement is transparent
- Validation remains continuously relevant
- Documentation reflects operational reality
Training records, access controls, and vendor activities are governed within a single oversight framework. When inspectors ask how something occurred, the system answers without interpretation.
Rather than relying on corrective preparation before inspections, organisations operate in a constant state of readiness.Inspection becomes confirmation, not investigation.
Why Silent Risks Matter More Than Visible Failures
Visible failures are usually corrected quickly. Silent compliance risks are more dangerous because they erode regulatory confidence. Once inspectors identify loss of control, scrutiny intensifies across systems, processes, and partners.
Preventing these risks protects more than compliance.
It protects :
- Organisational credibility
- Regulatory relationships
- Long-term operational stability
Conclusion
Silent compliance risks are not operational mistakes. They are design failures. They emerge when systems are built to deliver outcomes but not to explain them.
At Fidelity Health Services, PV-IT architectures are designed to make compliance demonstrable at every step. Through structured system design, continuous validation, and integrated oversight, silent risks are identified before inspections begin.
In modern pharmacovigilance, readiness is not proven on inspection day. It has been proven every day before.
Explore related insights in Compliance and Inspection Readiness in Pharmacovigilance and Safety System Modernisation — Building the Infrastructure of Compliance.