February 02, 2026
Pharmacovigilance Readiness for Mergers, Acquisitions, and Licensing
In mergers, acquisitions, and licensing deals, pharmacovigilance is rarely the headline topic. Yet it is one of the most common reasons deals slow down, lose value, or require post-transaction remediation.

PV risks are often invisible until diligence begins. Safety data appears complete. Reports are submitted. Vendors are in place. But once reviewers ask how systems are validated, how data lineage is preserved, and who retains regulatory control, weaknesses emerge.
Pharmacovigilance readiness for transactions is not about producing documents quickly. It is about proving that safety operations are stable, transferable, and regulator-defensible under scrutiny.
Why PV Becomes a Deal Risk?
Unlike finance or manufacturing, pharmacovigilance obligations cannot be paused or renegotiated after a transaction closes. Regulatory accountability transfers immediately, regardless of system maturity or vendor arrangements.
During diligence, acquirers and partners look for evidence that safety operations can continue without disruption. If PV systems rely heavily on informal practices, undocumented controls, or fragmented vendors, risk premiums increase.
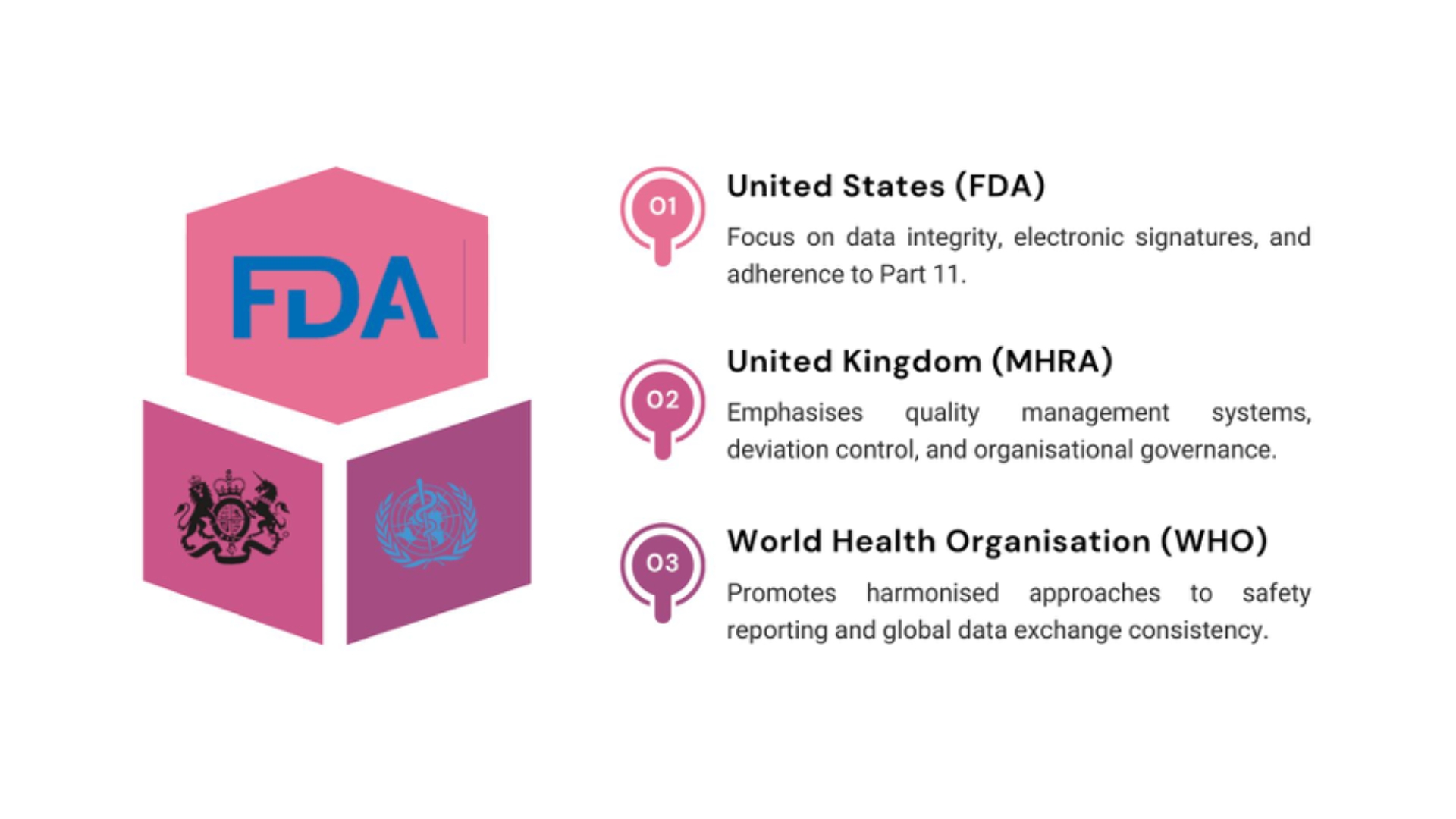
Regulators such as the European Medicines Agency and the U.S. Food and Drug Administration do not recognise transaction complexity as a justification for non-compliance. Responsibility is absolute from day one.
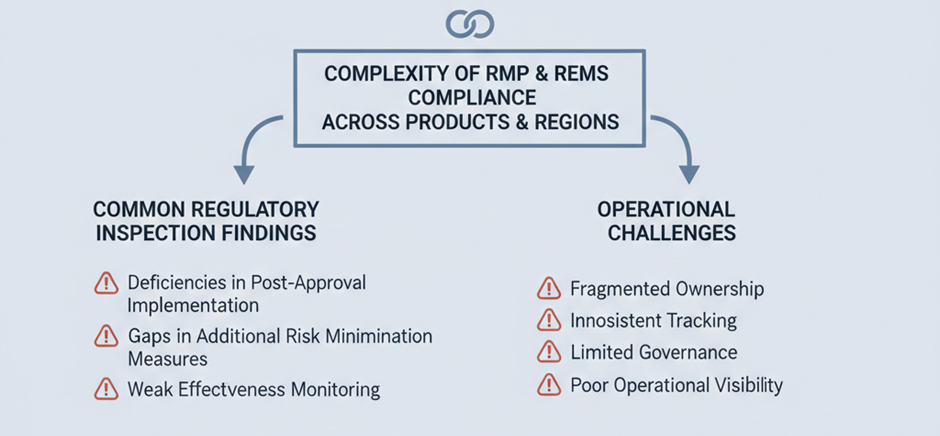
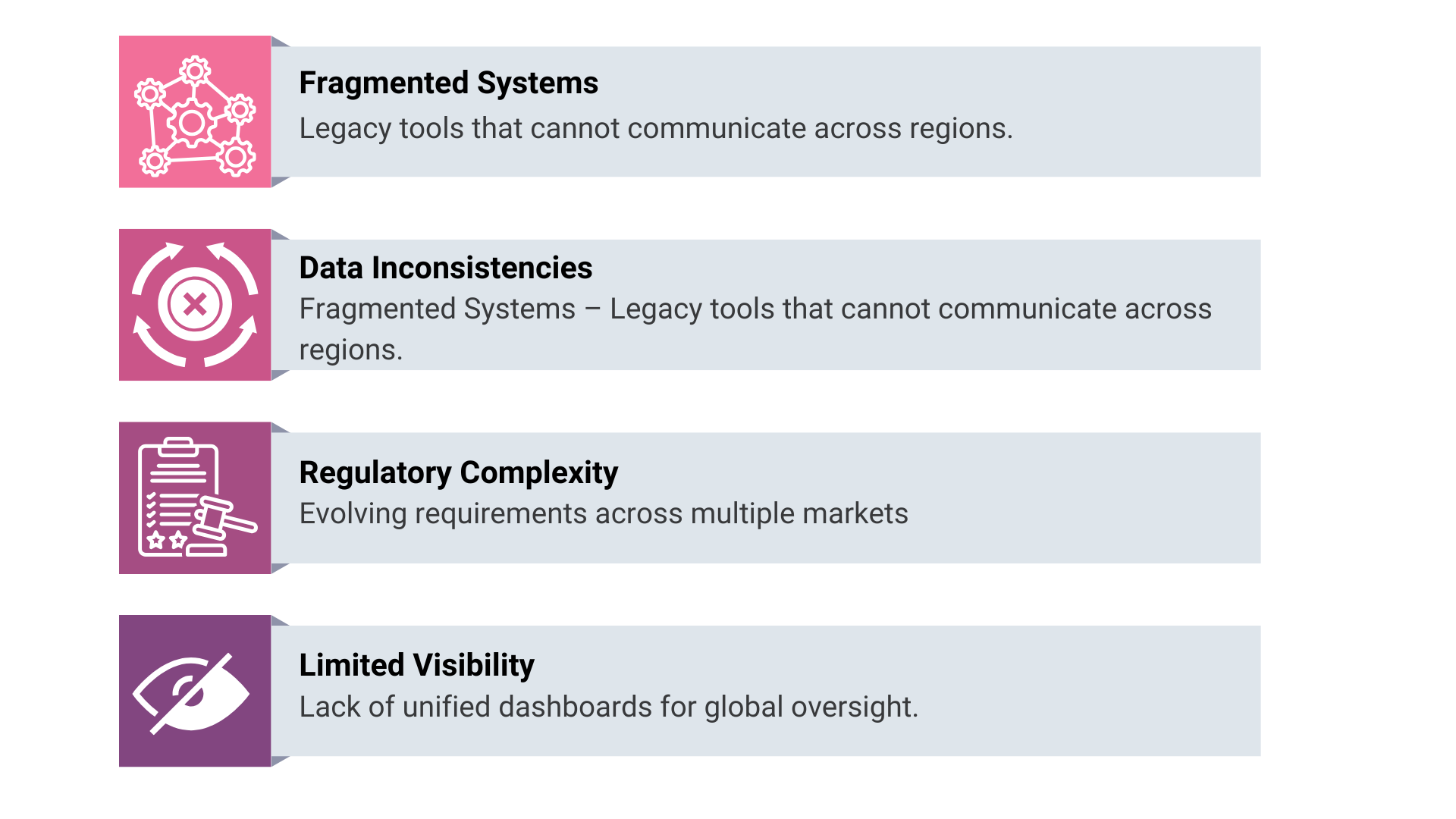
Where PV Due Diligence Commonly Fails?
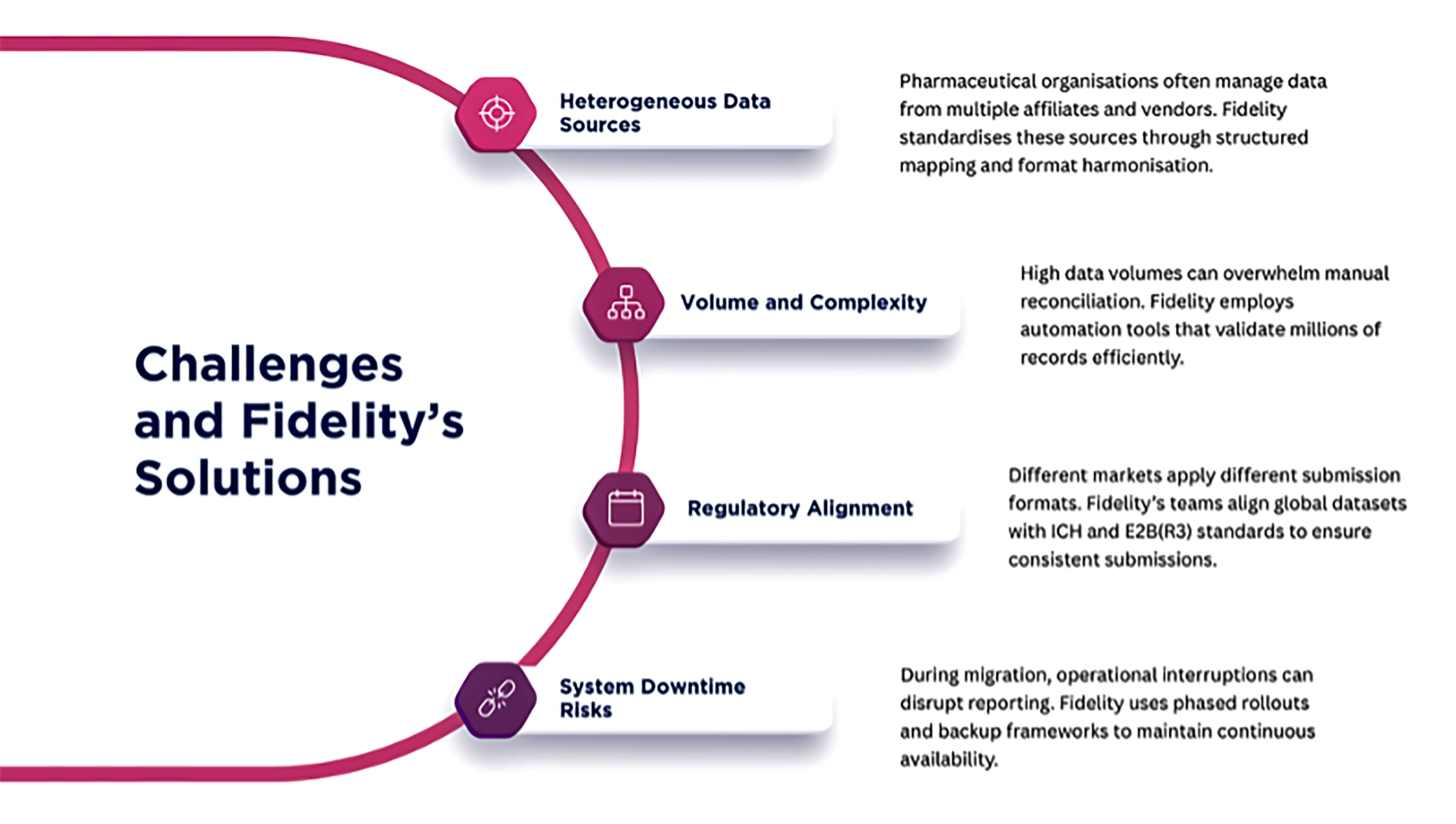
One of the first red flags appears when safety data ownership is unclear. In many organisations, data is distributed across sponsors, CROs, and technology vendors. If the sponsor cannot demonstrate immediate access and control, continuity risk is assumed.
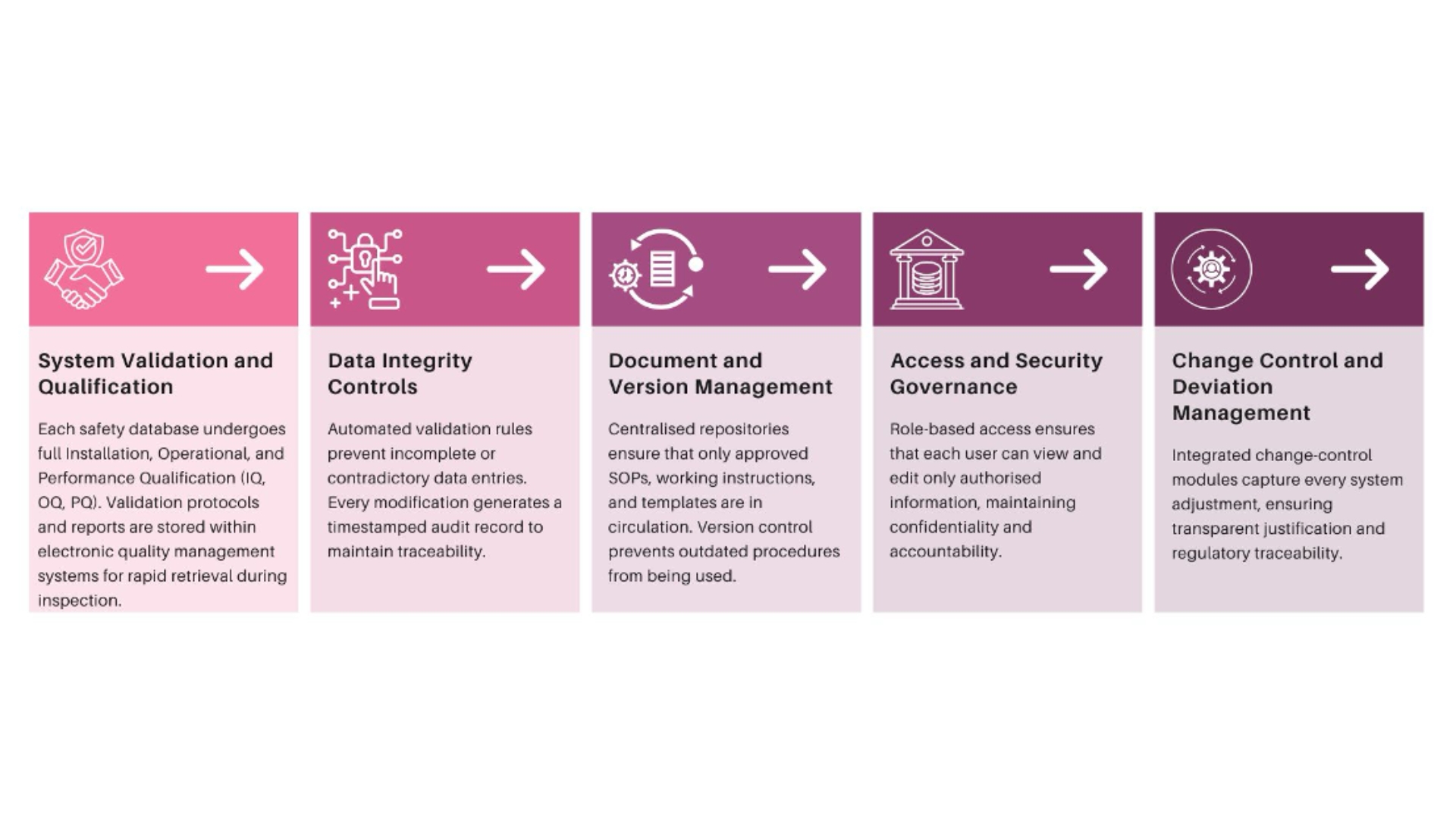
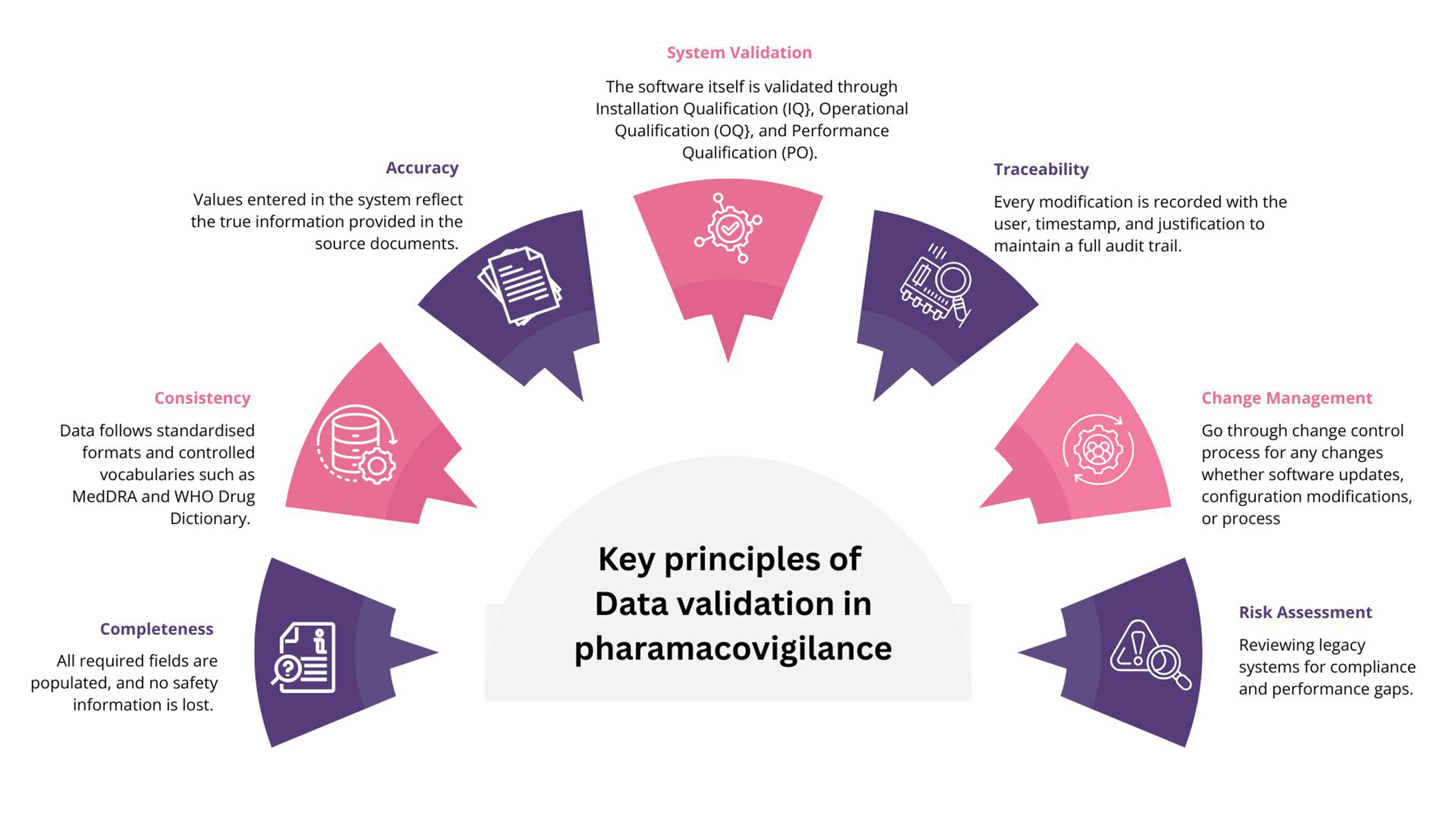
Validation gaps also surface quickly. Systems may have been validated historically, but diligence teams assess whether validation evidence aligns with current configurations. When migrations, upgrades, or vendor transitions lack complete documentation, confidence erodes.

Another common issue involves role definition. Licensing and co-development agreements often divide PV responsibilities across parties. If RACI models are not operationalised within systems, reviewers question whether obligations can be executed consistently.
Training and access governance are also scrutinised. Acquirers verify whether qualified personnel performed regulated activities and whether permissions reflect current responsibilities. Gaps here signal weak operational control.
Why Transaction Timelines Expose Hidden Weaknesses?
Transactions compress time. What normally unfolds gradually during inspections is accelerated during diligence. Reviewers do not evaluate intent. They evaluate evidence.
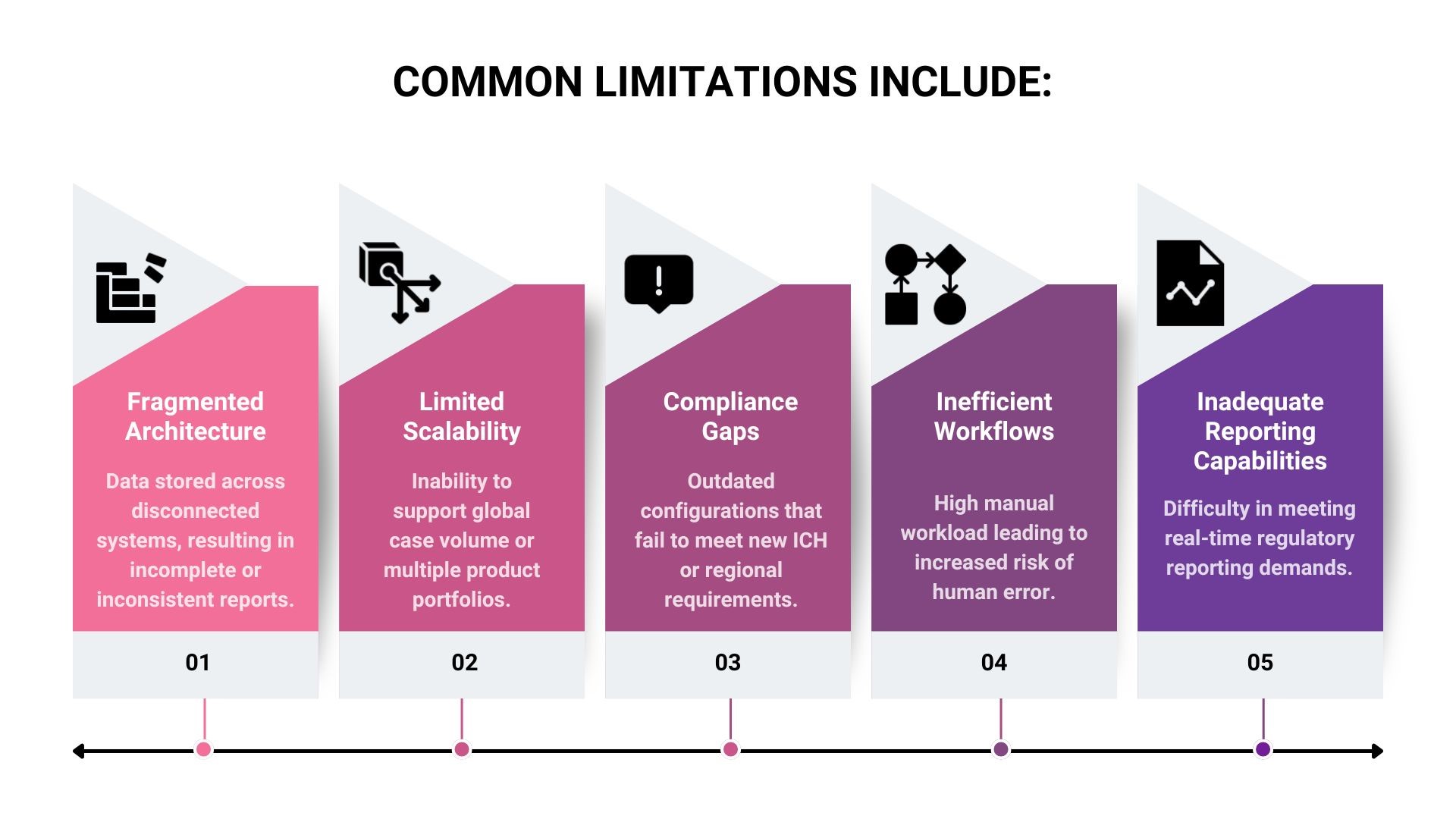
Organisations that operate comfortably day to day may struggle to answer basic questions when required to demonstrate controls immediately. This is where silent compliance risks transform into valuation concerns.
Without inspection-ready PV-IT infrastructure, teams rely on manual explanations rather than system proof. In diligence environments, explanations are rarely sufficient.
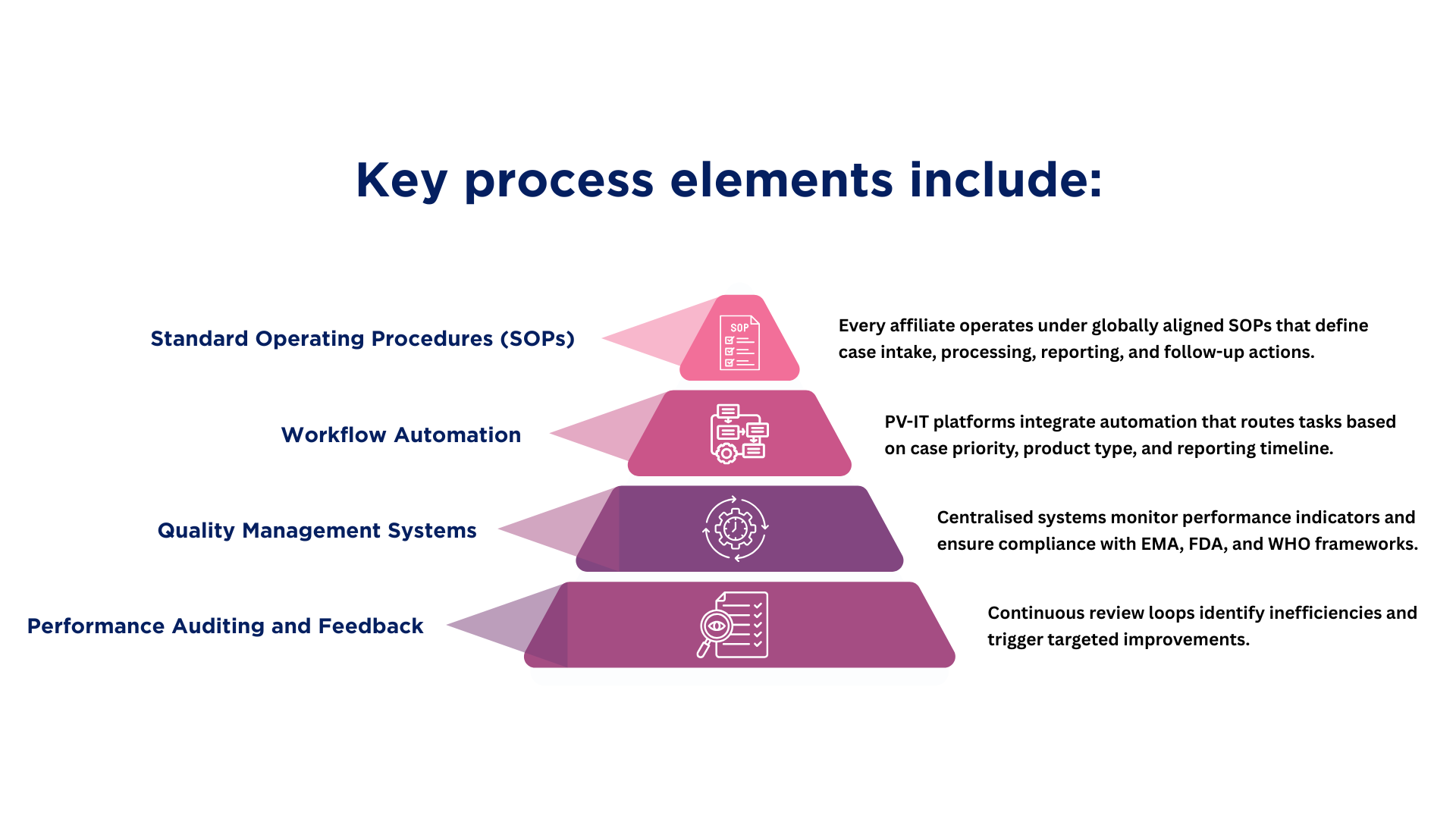
Designing PV for Transaction Readiness
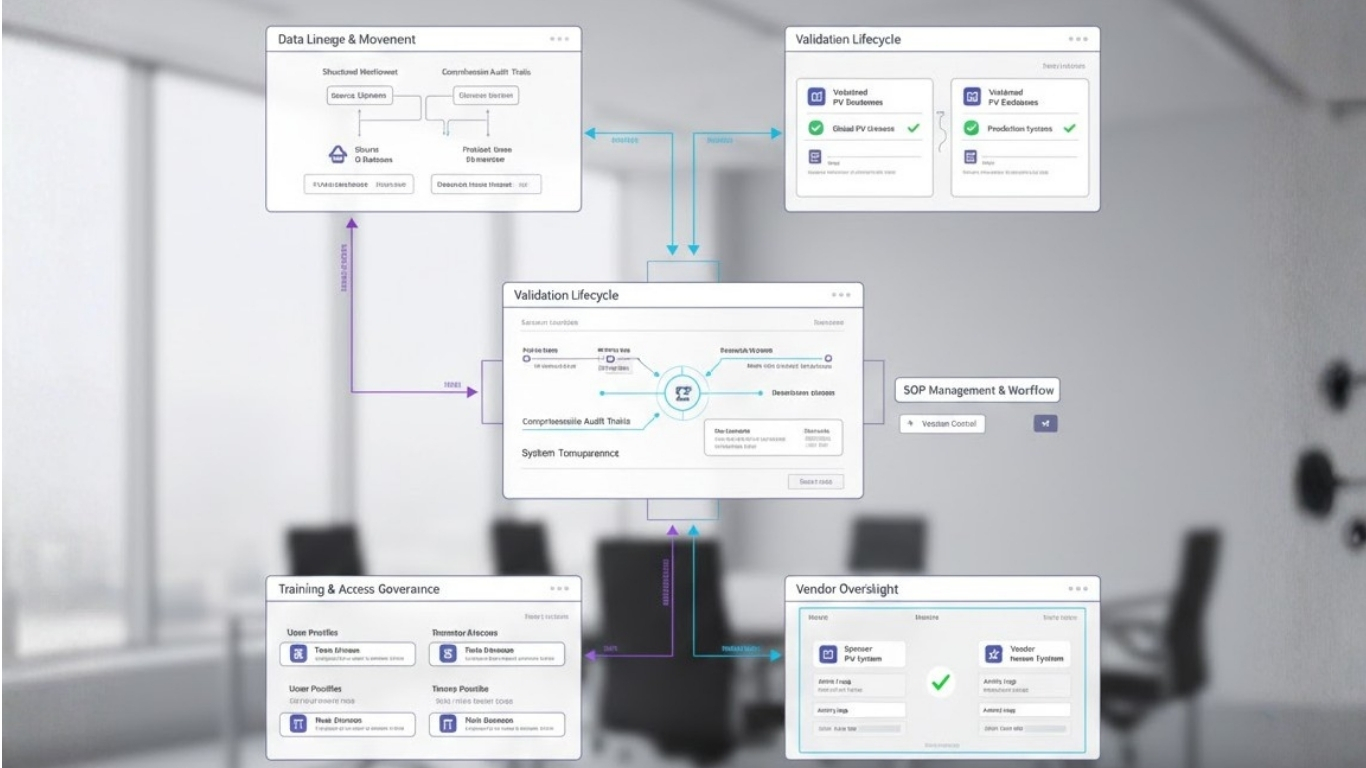
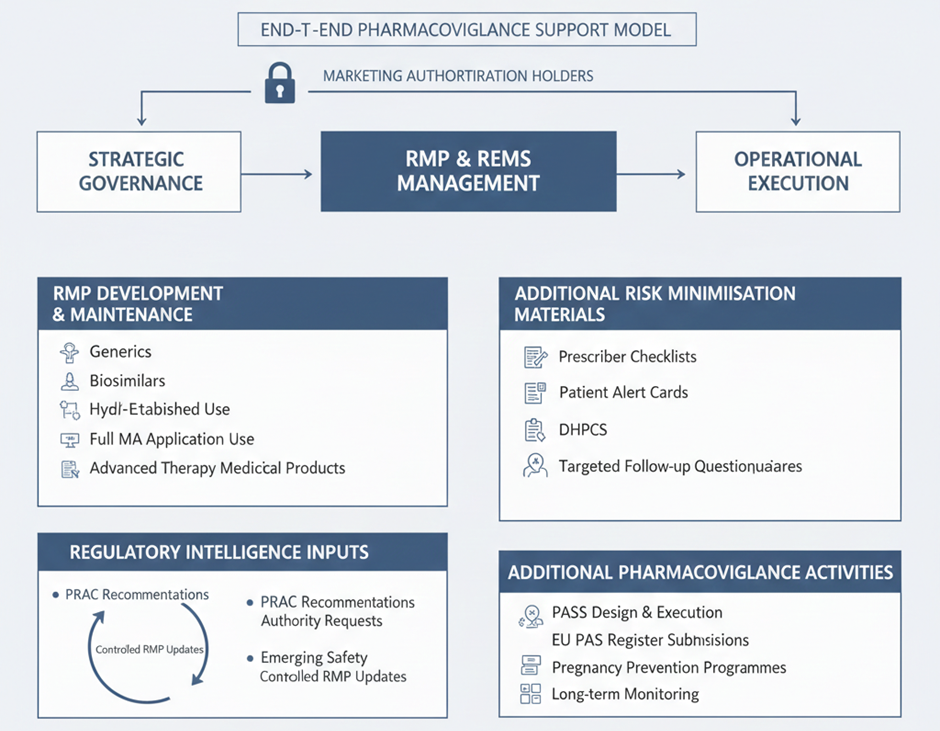
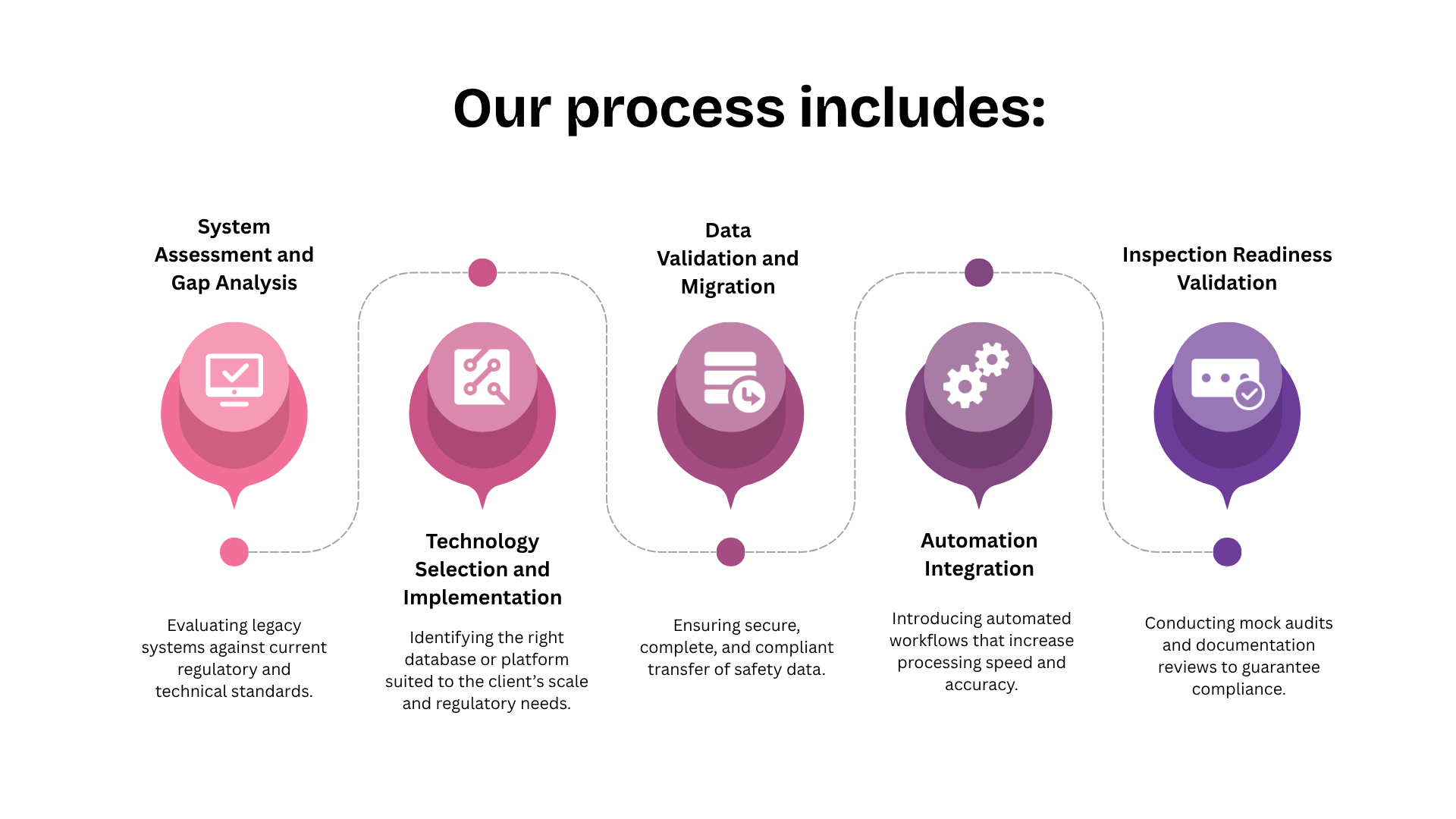
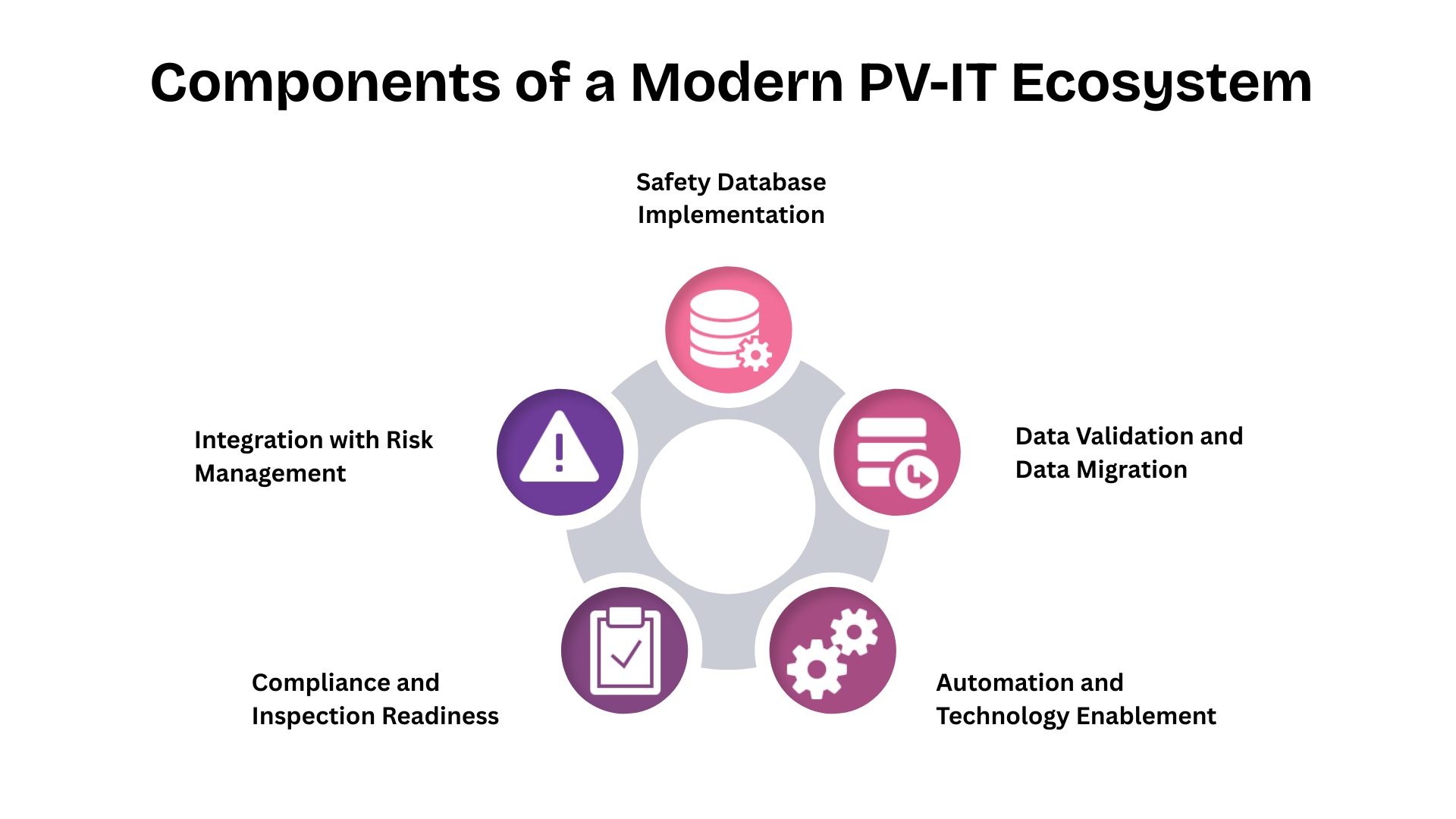
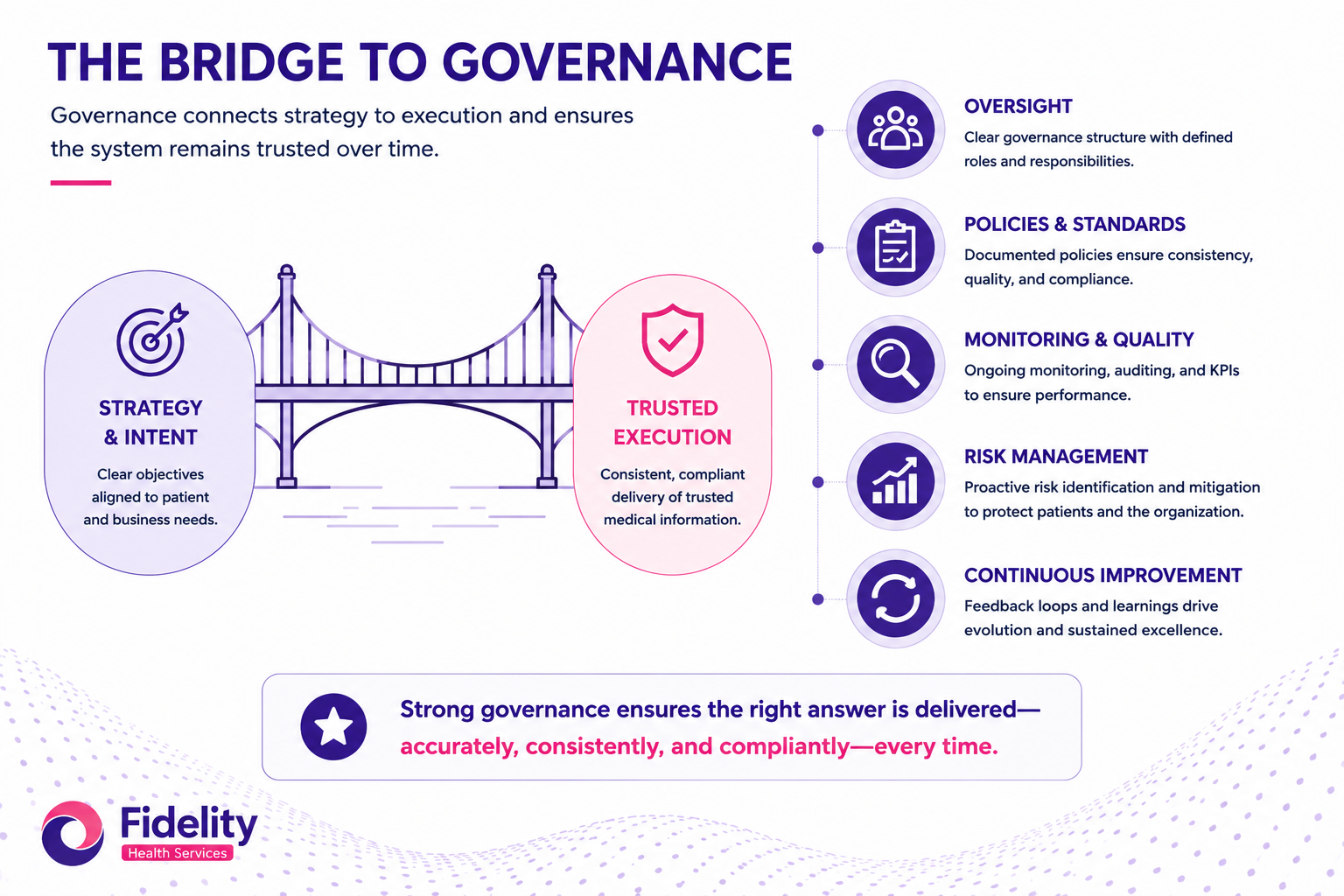
At Fidelity Health Services, transaction readiness is treated as an extension of inspection readiness. Systems are designed to support continuity across ownership and operational models.
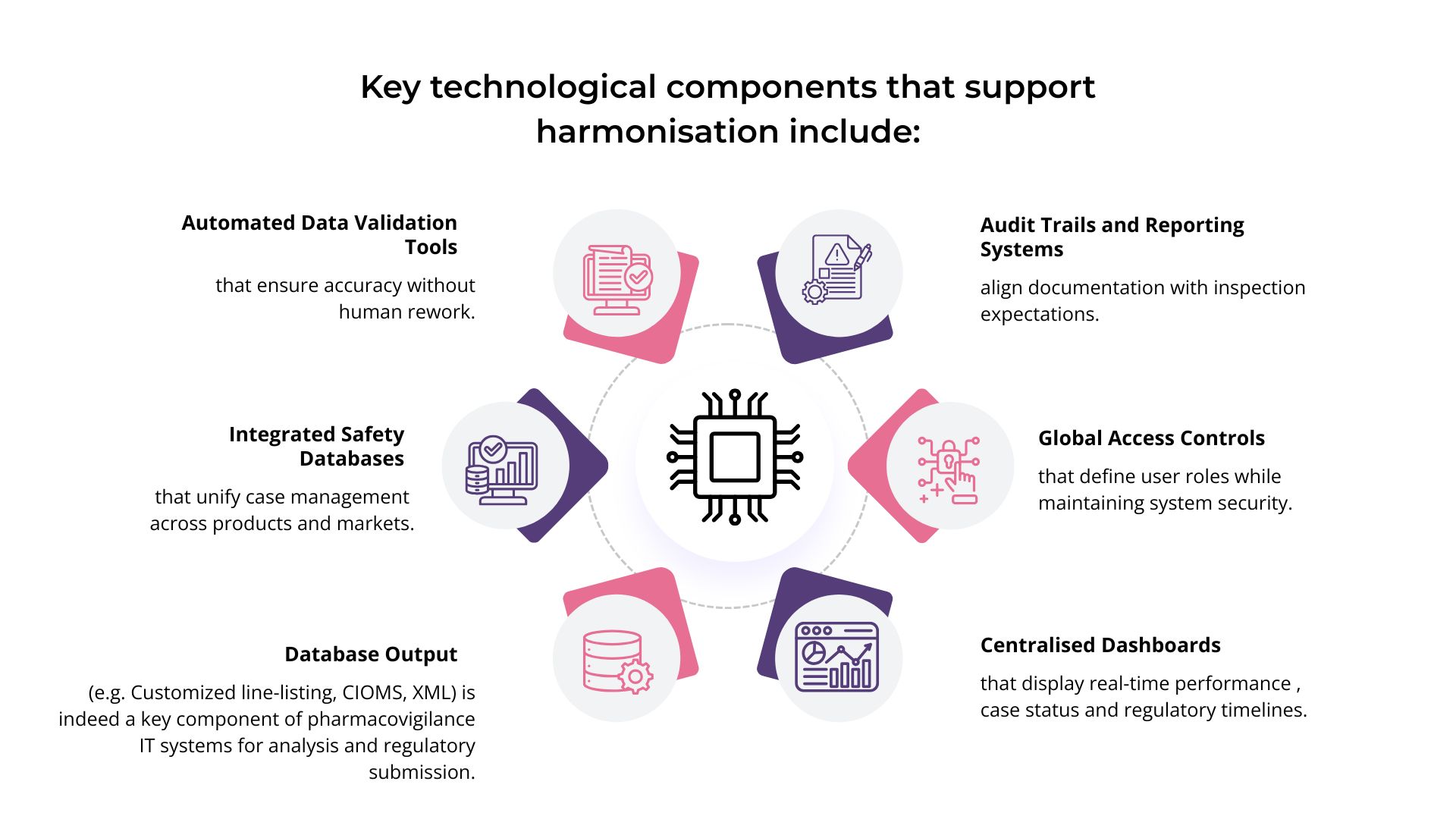
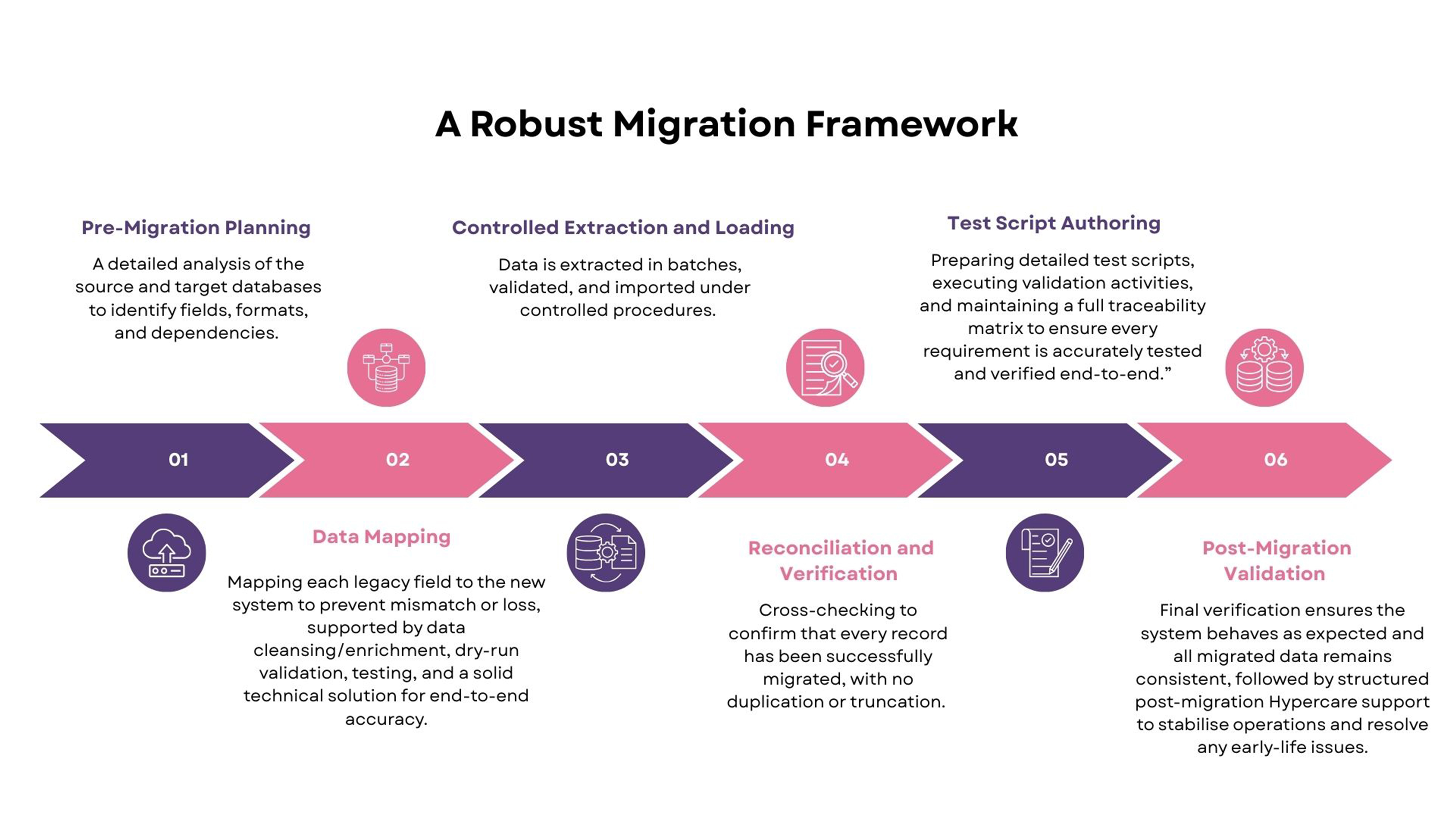
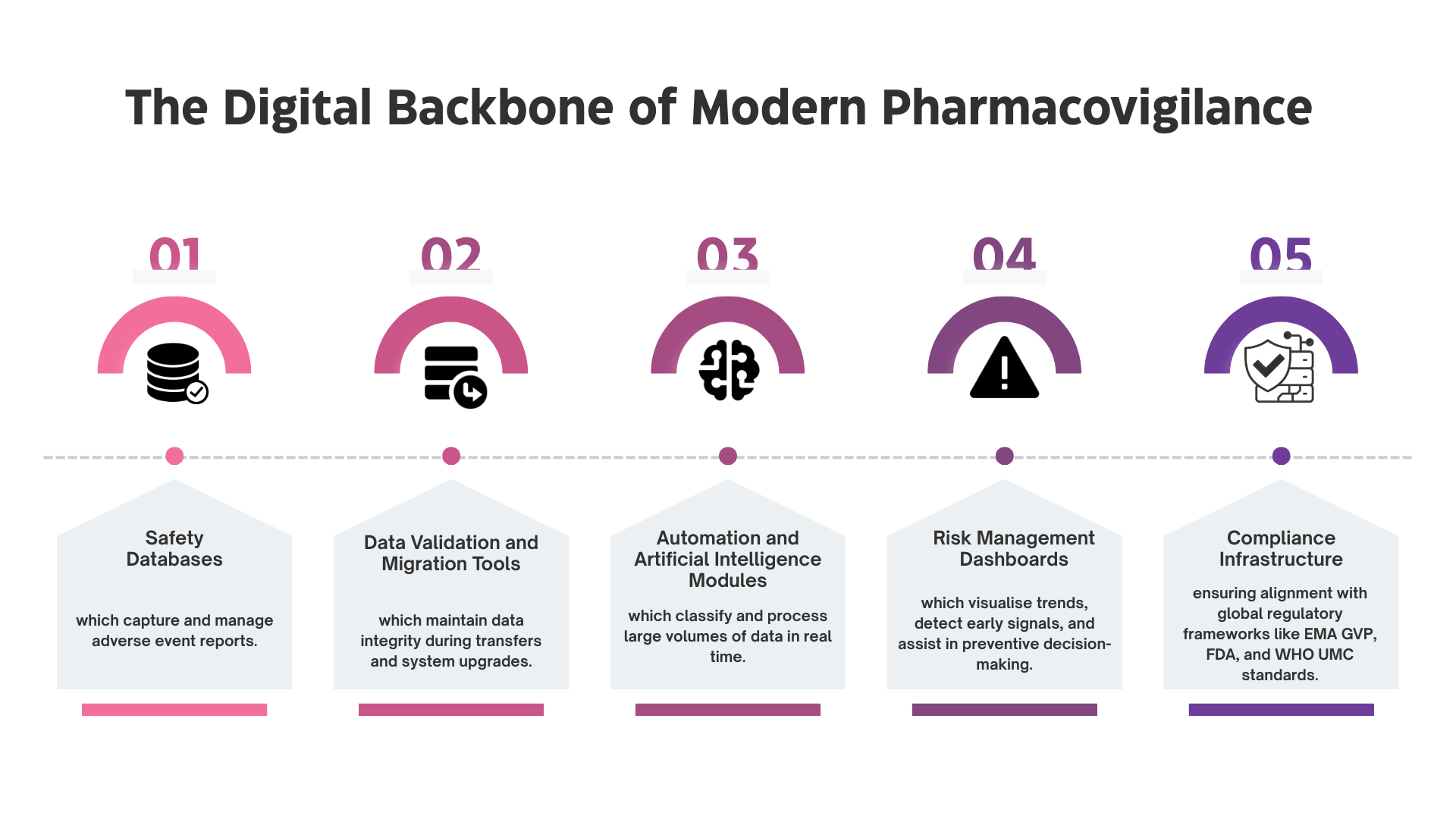
PV-IT environments are structured to ensure that data lineage remains intact regardless of sponsor or vendor changes. Validation evidence is centralised and continuously updated, allowing rapid demonstration of system integrity.

Governance models are embedded within workflows so that role transitions can be mapped, audited, and approved without operational disruption. Training records and access controls remain synchronised as responsibilities shift.
This approach allows pharmacovigilance operations to remain stable even as corporate structures change.
Licensing and Co-Development Considerations
Licensing arrangements introduce additional complexity. Safety responsibilities are often shared, yet regulators hold each marketing authorisation holder fully accountable.
Effective PV-IT design supports shared visibility without shared ambiguity. Each party can demonstrate its role, oversight activities, and decision authority within a unified system framework.
This clarity reduces inspection risk and prevents disputes post-transaction, when accountability becomes critical.
Regulatory Perspective on Transactional PV
Global authorities expect pharmacovigilance systems to remain compliant regardless of organisational change. Guidance from the International Council for Harmonisation emphasises continuity of safety oversight across the product lifecycle.

The World Health Organisation similarly promotes harmonised safety systems capable of sustaining transitions without data loss or control gaps.
Transaction readiness is therefore not optional. It is a regulatory expectation.
Why PV Readiness Protects Deal Value?
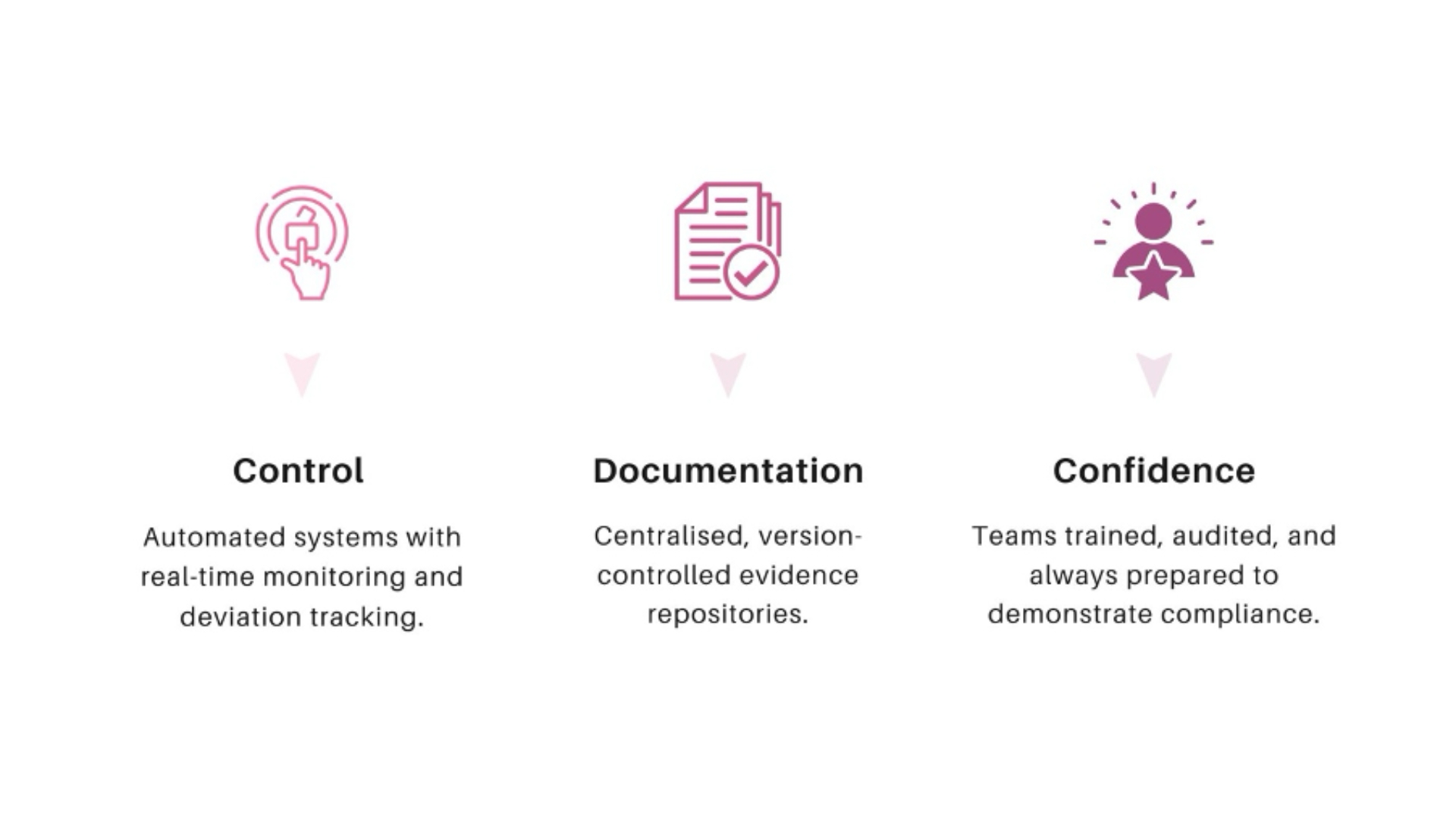
When pharmacovigilance systems are inspection-ready, diligence becomes verification rather than investigation. Questions are answered with evidence. Risks are quantified rather than assumed.
This reduces remediation costs, accelerates closing timelines, and strengthens trust between partners. In competitive deal environments, PV readiness can become a differentiator rather than a liability.
Conclusion
Pharmacovigilance is not a background function in mergers, acquisitions, and licensing. It is a core determinant of regulatory continuity and deal confidence.
At Fidelity Health Services, PV-IT frameworks are designed to withstand both inspections and transactions. By embedding traceability, validation, and governance into system design, pharmacovigilance operations remain stable through change.
In a market where deals move quickly and regulators do not pause, readiness is not preparation. It is architecture.
Explore related insights in Compliance and Inspection Readiness in Pharmacovigilance and Operational Harmonisation in Pharmacovigilance.










 service model.jpg)